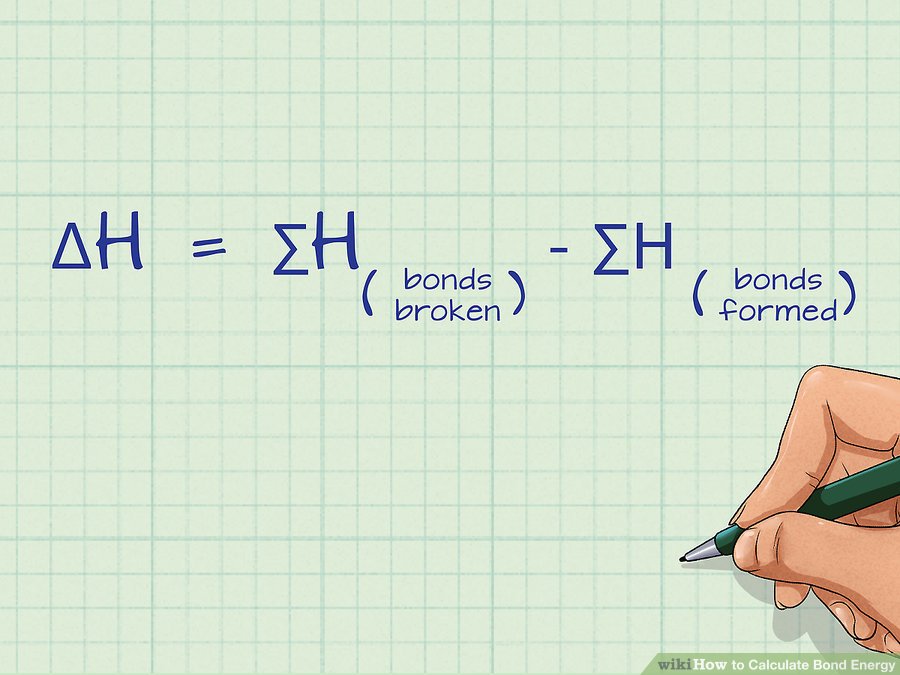Unit 6 Thermochemistry & Unit 9 Thermodynamics
Using Gibb’s Free Energy - Bozeman Science Video #59 (7:56)
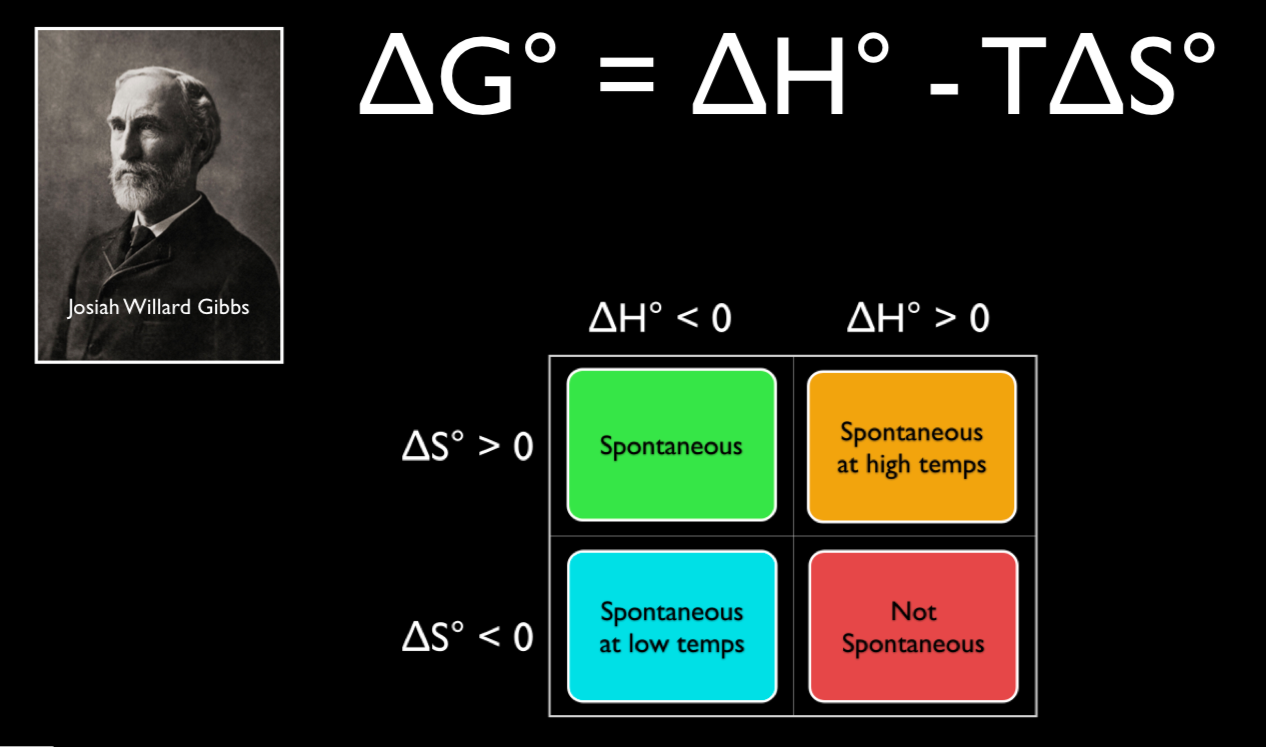
In this video Paul Andersen explains how you can use the Gibbs Free Energy equation to determine if a process is spontaneous or not spontaneous. If the ΔG is less than zero the process is spontaneous. If the ΔG is greater than zero the process is not spontaneous.
In this video Paul Andersen explains how you can use the Gibbs Free Energy equation to determine if a process is spontaneous or not spontaneous. If the ΔG is less than zero the process is spontaneous. If the ΔG is greater than zero the process is not spontaneous.
HEAT FLOWS DOWNHILL... What do you have to say about that?
Question: What is a simple defintion of the laws of thermodynamics?
Thermodynamics is the study of the inter-relation between heat, work and internal energy of a system.
The British scientist and author C.P. Snow had an excellent way of remembering the three laws:
Answered by: Dan Summons, Physics Undergrad Student, UOS, SouhamptonIn simplest terms, the Laws of Thermodynamics dictate the specifics for the movement of heat and work. Basically, the First Law of Thermodynamics is a statement of the conservation of energy - the Second Law is a statement about the direction of that conservation - and the Third Law is a statement about reaching Absolute Zero (0� K).
However, since their conception, these laws have become some of the most important laws of all science - and are often associated with concepts far beyond what is directly stated in the wording. To give you a better understanding on how these laws came about and their modern scope of coverage, you have to understand when and why these laws were generated.
Read the rest of the story here.
Thermodynamics is the study of the inter-relation between heat, work and internal energy of a system.
The British scientist and author C.P. Snow had an excellent way of remembering the three laws:
- You cannot win (that is, you cannot get something for nothing, because matter and energy are conserved).
- You cannot break even (you cannot return to the same energy state, because there is always an increase in disorder; entropy always increases).
- You cannot get out of the game (because absolute zero is unattainable).
Answered by: Dan Summons, Physics Undergrad Student, UOS, SouhamptonIn simplest terms, the Laws of Thermodynamics dictate the specifics for the movement of heat and work. Basically, the First Law of Thermodynamics is a statement of the conservation of energy - the Second Law is a statement about the direction of that conservation - and the Third Law is a statement about reaching Absolute Zero (0� K).
However, since their conception, these laws have become some of the most important laws of all science - and are often associated with concepts far beyond what is directly stated in the wording. To give you a better understanding on how these laws came about and their modern scope of coverage, you have to understand when and why these laws were generated.
Read the rest of the story here.
BIG IDEA 5
Thermodynamics describes the role energy plays in physical and chemical changes.
|
Bozeman Videos
Big Idea 5 -Thermodynamics
Bozeman for AP Chemistry |
Other Videos
Temperature
Misconceptions About Temperature (3:59) Veritasium
Thermochemistry
Intro to Thermochemistry by JRF Science (6:34)
Mr. Key introduces the concept of evaluating energy flow as a part of chemical reactions, the study of which is referred to as thermochemistry Calorimetry - Tyler DeWitt
Calorimetry - Teacher's Pet
Calorimetry
Learn about calorimetry, heat, enthalpy and calorimetry calculations to find specific heat in this video! Teacher's Pet (4:52) Thermodynamics & Enthalpy
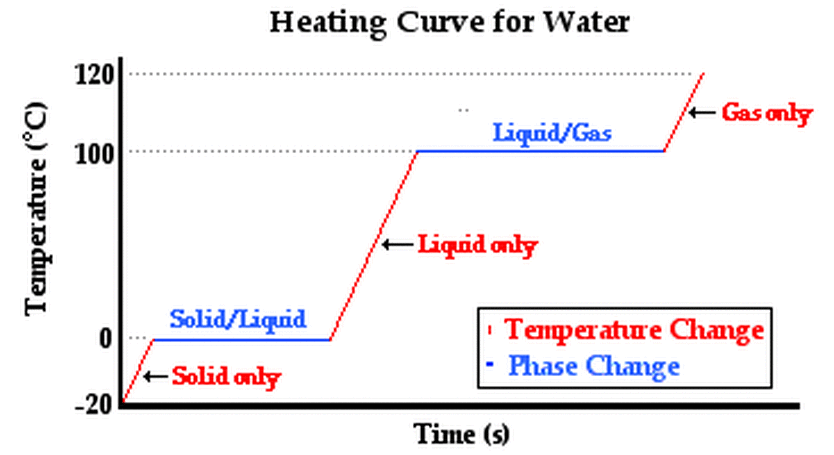
Heating Curves
Heat in Changes of State
Learn about energy diagrams, calculating molar heat of vaporization and fusion and molar heat of solution in this video! Teacher's Pet (6:56) Hess's Law
|
Practice
Worksheets
Bond Enthalpy (Bond Energy)
|
Other
Student Guides from your Text
AP College Board Curriculum Framework
Helpful PowerPoints
Lab Resources
Simulations
Explore how heating and cooling iron, brick, and water adds or removes energy. See how energy is transferred between objects. Build your own system, with energy sources, changers, and users. Track and visualize how energy flows and changes through your system.
|
Specific Heat Capacity
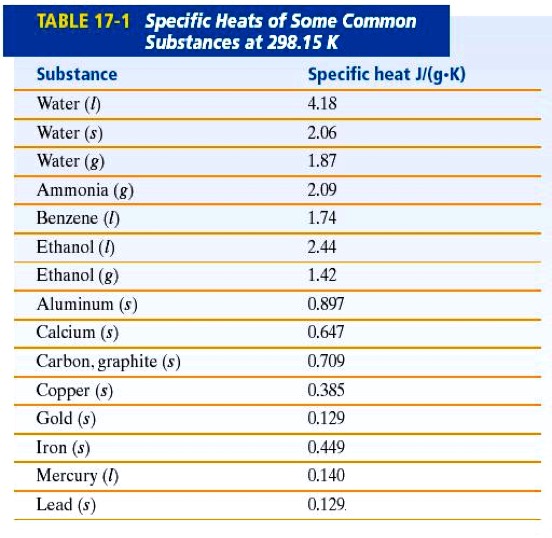
Specific Heat Capacity (Cp) - The quantity of heat required to raise the temperature 1 gram of a substance by one degree Celsius is called the specific heat capacity of the substance. The quantity of heat is frequently measured in units of Joules (J).
Specific Heat
Another property, the specific heat (C) (sub S), is the heat capacity of the substance per gram of the substance.
The specific heat of water is 4.18 J/g° C.
The specific heat of water is 4.18 J/g° C.
Which of the substances on this chart has the highest Specific Heat?
q = m x C x (Tf - Ti)
q = quantity of heat energy gained or lost by substance (Joules or calories)
m = mass of sample (grams)
C = heat capacity (J oC-1 g-1 or J K-1 g-1)
Tf = final temperature
Ti = initial temperature
m = mass of sample (grams)
C = heat capacity (J oC-1 g-1 or J K-1 g-1)
Tf = final temperature
Ti = initial temperature
Thermodynamics
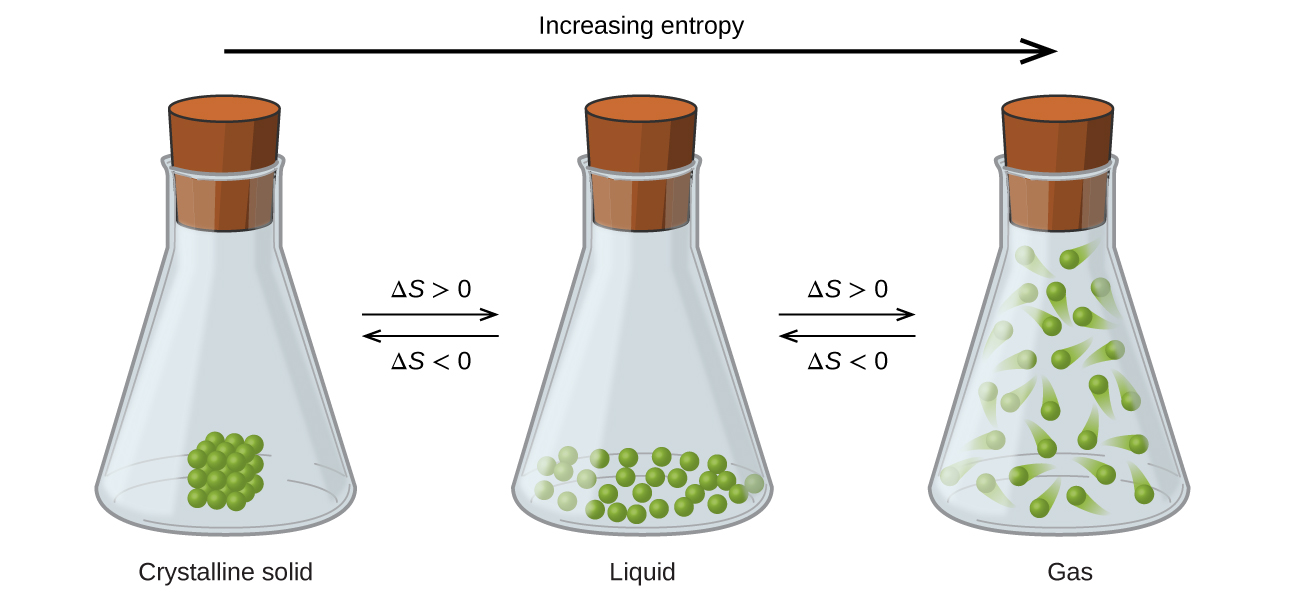
Entropy (S)
Entropy - In thermodynamics, entropy (usual symbol S) is a measure of the number of specific realizations or microstates that may realize a thermodynamic system in a defined state specified by macroscopic variables. Most understand entropy as a measure of molecular disorder within a macroscopic system. The second law of thermodynamics states that an isolated system's entropy never decreases.
Make sure that you know the difference between the two ways to calculate ΔH from "data given in a table."
ΔH = (sum of enthalpies of formation for the products) - (sum of the enthalpies of formation for the reactants)
ΔH = (sum of bond enthalpies for the bonds that are broken) - (sum of bond enthalpies for the bonds that are formed)
I mention this because on the 2017 exam, in FRQ #2(b), a lot of students lost a point for confusion between these two methods. (See two examples of student responses in the picture.)
ΔH = (sum of enthalpies of formation for the products) - (sum of the enthalpies of formation for the reactants)
ΔH = (sum of bond enthalpies for the bonds that are broken) - (sum of bond enthalpies for the bonds that are formed)
I mention this because on the 2017 exam, in FRQ #2(b), a lot of students lost a point for confusion between these two methods. (See two examples of student responses in the picture.)
There is no other quantitative variable in AP chemistry that shows up in so many different problems as ΔH.
You need to be able and calculate ΔH in at least 5 different ways:
1) From Heats of Formation tables,
2) Hess's Law
3) From Bond Energy tables,
4) From experimental heat/q values divided by moles,
5) Solving the Gibbs Free Energy Equation.
You need to be able and calculate ΔH in at least 5 different ways:
1) From Heats of Formation tables,
2) Hess's Law
3) From Bond Energy tables,
4) From experimental heat/q values divided by moles,
5) Solving the Gibbs Free Energy Equation.
How to Calculate Bond Energy
FLASHCARDS to Satisfy Your Compulsion For Repetition and Memorization!