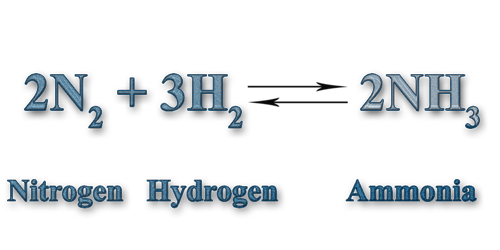Unit 13 General & Solubility Equilibria
While we know chemical equilbrium is no longer the topic of Question 1 of the FR, we DO know that is still important enough to have its own Big Idea! (Big Idea 6) We have also learned that it is best to learn Equilbrium in two parts: Unit 13 - General Equilibrium: Kc, Kp and Ksp & Unit 14 - Acid-Base Equilibrium and Buffers.
The Haber Process - Turning air into fertilizer and doubling food production since 1908.
From the TED-Ed Lesson The chemical reaction that feeds the world - Daniel D. Dulek
Animation by Uphill Downhill
TED-Ed Video The Chemical Reaction That Feeds The World (5:18) [here]
From the TED-Ed Lesson The chemical reaction that feeds the world - Daniel D. Dulek
Animation by Uphill Downhill
TED-Ed Video The Chemical Reaction That Feeds The World (5:18) [here]
|
Video
|
Games/Worksheets/Practice
|
Other Resources
|
|
Le Chatelier’s Principle
Le Chatelier's Principle Lab with Cobalt Complex Ions (10:09) [here]
Le Chatelier's Principle - Professor Dave Explains (4:08) [here] If a system is at equilibrium, and we do something to it, it will shift in a particular way. It is quite easy to predict the behavior of equilibria if we know about Le Chatelier's principle and three simple situations!
RICE Tables
Equilibrium Equations: Crash Course Chemistry #29
Hank shows you that, while it may seem like the Universe is messing with us, equilibrium isn't a cosmic trick. Here, he shows you how to calculate equilibrium constant & conditions of reactions and use RICE tables all with some very easy, not-so-scary math. ICE Tables made EASY! ~Justin Mueller (7:53) [here]
How To Solve Quadratic Equations Using The Quadratic Formula ~The Organic Chemistry Tutor (5:55) [here]
The Haber Process
The Haber Process: Learn the basics about Ammonia and how it is used in our every day life and in industry. (3:54) [here]
Fritz Haber - Great Minds
~SciShow (9:40) Brilliant and heartless Fritz Haber, a great mind who is considered "the father chemical warfare," but who also made discoveries and innovations that helped lead to the Green Revolution which is credited with preventing the starvation of over a billion people. [here] Fritz Haber - Great or Evil Read the short story [here]
How Do You Solve a Problem Like Frizt Haber? Radio Lab, the NPR science program, did a show on Fritz Haber that sent chills down my spine when I heard it. You can listen to it [here]
Haber-The Father of Chemical Warfare (The Movie Link) ~ChemEdX [here]
Copper II with Ammonia
Complex ions are used as indicators in chemical reactions, and even in the body. The body uses complexes with iron and oxygen to cary the oxygen to locations in the body. (1:43) [here] |
The Haber Process
TedEd Lesson The Chemical Reaction That Feeds The World [here]
Equilibrium Concentrations
Calculating Equilibrium Concentrations Science Geek [here]
Calculating K
Calculating K the Equilibrium Constant - Science Geek [here]
Kc and Kp
The Relationship Between Kc and Kp - Science Geek [here]
Evaluating Reaction Direction, Q and K
Working with Q and K - Science Geek [here]
Le Chatelier’s Principle
Le Chatelier’s Principle Practice Problems - Instant Feedback from Science Geek [here]
Solubility Product Constant Ksp
|
Forest Classroom Resources:
Helpful PowerPoints
|
|
Bozeman Videos
062 - Reversible Reactions (5:39) [here]
063 - The Reaction Quotient (7:08) [here]
064 - Equilibrium (12:24) [here]
065 - The Equilibrium Constant (6:14) [here]
066 - LeChatelier’s Principle (7:00) [here]
067 - Equilibrium Disturbances (5:36) [here]
068 - Acid-Base Equilibrium (10:26) [here]
069 - pH & Buffers (5:56) [here]
070 - Solubility (7:05) [here]
071 - Free Energy & the Equilibrium Constant (6:44) [here]
|
Dr. Dena Legget’s Videos
Equilibrium 1 - Introduction (11:38) [here]
Equilibrium 2 - Law of Mass Action (14:25) [here]
Equilibrium 3 - Impacts of Manipulations on K (16:30) [here]
Equilibrium 4 - RICE (11:30) [here]
Equilibrium 5 - Let's Do It (7 & 8) [here]
Equilibrium 6 -Let's Do It 9 & 10 [here]
Equilibrium 7 - Ksp Solubility Constant [here]
Equilibrium 8 -Ksp Let's Do It Exercises [here]
Equilibrium 9 - Common Ion Effect [here]
Equilibrium 10 - Le Chatelier's Principle [here]
Equilibrium 11 - Le Chatlier's Principle Cont. [here]
Equilibrium 12 - van't Hoff (not on AP test) [here]
|
Equilibrium Quizlet
Speaking of Chemical Warfare...How a simple method could help save people from nerve gas attacks [here]
Learning Outcomes After studying this chapter, you should be able to:
- Explain what is meant by chemical equilibrium and how it relates to reactions rates.
- Write the equilibrium-constant expression for any reaction.
- Given the value of Kc convert to Kp and vice versa.
- Relate the magnitude of an equilibrium constant to the relative amounts of reactants and products present in an equilibrium mixture.
- Manipulate the equilibrium constant to reflect changes in the chemical equation.
- Write the equilibrium-constant expression for a heterogeneous reaction.
- Calculate an equilibrium constant from concentration measurements.
- Predict the direction of a reaction given the equilibrium constant and the concentrations of reactants and products.
- Calculate the equilibrium concentrations given the equilibrium constant and all but one equilibrium concentration.
- Calculate equilibrium concentrations, given the equilibrium constant and the starting concentrations.
- Use Le Chatelier’s principle to predict how changing the concentrations, volume, or temperature of a system at equilibrium affects the equilibrium position.
General Equilibrium Quizlett
|
Haber Process Animation by Uphill Downhill
|