Unit 1 - Introduction to Chemistry
Unit 1 Part One: Chemistry and The Scientific Method
Learning Targets: By the end of the unit, a student in this course should be able to...
- Describe chemistry in a general sense.
- Identify the five areas of chemistry.
- Explain why the study of chemistry is important.
- Explain how chemists impact multiple studies.
- Work in a lab setting following published safety guidelines.
- Use the steps of the scientific method to develop an experiment using the OAIM (Object, Action, Instrument, Measurement) method.
- Answer a proposed question with experimental data using CER (Claim, Evidence, Reasoning).
- Complete a lab write up using the Four Corner method.
Vocabulary
|
|
|
|
Unit 1 Part Two: Measurement and Math for Chemistry
Learning Targets: By the end of the unit, a student in this course should be able to...
- evaluate the accuracy and precision of a set of measurements.
- report numbers to the correct number of significant figures, both measurements and calculations.
- express numbers in scientific notation and complete calculations in scientific notation WITHOUT a calculator.
- write measurements using the correct number of significant figures.
- identify the five SI base units commonly used in chemistry.
- work with measurements EXCLUSIVELY in the metric system, with emphasis on length, volume, mass, temperature, and energy.
- convert between similar units.
- perform dimensional analysis.
- experimentally determine the density of solids and liquids.
- identify an unknown substance using density.
- explain how temperature impacts density.
Vocabulary
|
|
|
|
Unit 1 Resources
|
Videos
Learning Chemistry
How to Learn Chemistry (and a Lesson on Collision Theory)
by Michael Farabaugh (6:43) Helpful advice for learning chemistry. Also includes a little about collision theory, which relates to chemical kinetics and reaction rates. [Here] Scientific Notation
Rearranging Equations
Significant Figures
Converting Units Using Dimensional Analysis
Converting Metric Units of Volume (9:33) ~Tyler DeWitt
Don't be lost... Learn how to convert.
Mixtures -
Separation Techniques |
Games/Practice
Scientific Notation
Significant Figures
|
Activities
Dimensional Analysis
Dr. Parkinson's Help Pages on Dimensional Analysis ~Southeastern Louisiana University [here]
Dimensional Analysis Worksheet with Answers [here]
Module 3: Calculating Medication Dosages - Practice Problems using Dimensional Analysis with Answers [here]
Dimensional Analysis for Nurses [here]
Significant Figures
Charts & Graphs
ANSWERS
|
Other
Class PowerPoints & Notes
Curriculum Documents
Significant Figures
Graphing
|
Metric Unit Names/Symbols Practice
Significant Figures
There are three rules on determining how many significant figures are in a number:
- Non-zero digits are always significant.
- Any zeros between two significant digits are significant.
- A final zero or trailing zeros in the decimal portion ONLY are significant
See the Full Explanation on Significant Figures Here
Good to Know
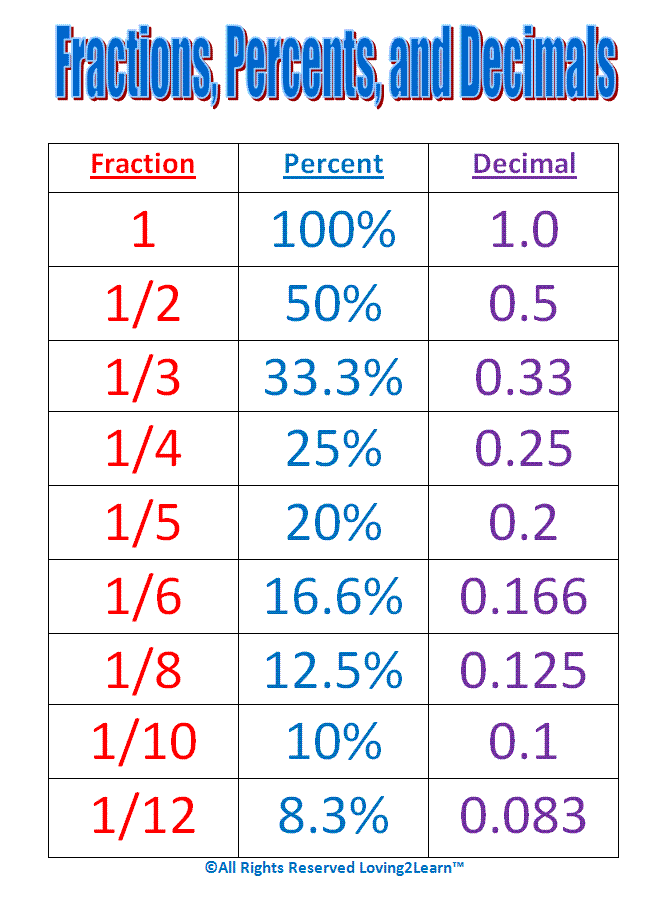
Memorize these Fractions and their Percents and Decimals: