Unit 2 - Chemical Names & Formulas
Learning Targets: By the end of the unit, a student in this course should be able to...
- I can understand that the drive for atoms to form bonds is based on the stability of the noble gases and the octet rule.
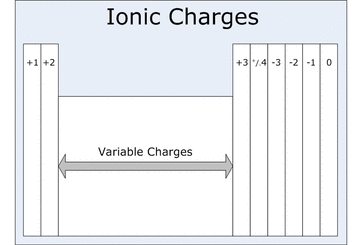
- I can describe the formation of an ionic bond.
- I can describe the formation of an anion or cation from its neutral atom.
- I can determine the correct ratio of cations to anions needed to form a neutral ionic compound.
- I can explain the difference between a monatomic and polyatomic ion.
- I can develop a flowchartthat can be used to name and write chemical formulas.
- I can state and apply the octet rule.
- I can understand that a formula unit represents one particle of an ionic compound.
- I can distinguish between ionic compounds and binary molecular compounds.
- I can name and write formulas for ionic compounds using IUPAC nomenclature (naming) rules.
- I can name and write formulas for binary molecular compounds.
Vocabulary
|
|
|
List of Polyatomic Ions
|
|
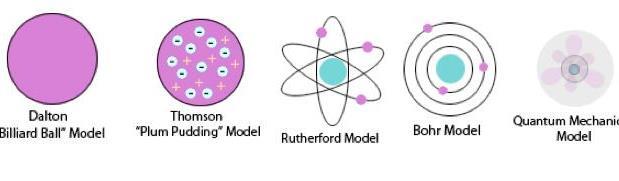
Videos History of the Atom
Isotopes
Find the Average Atomic Mass - Example: Magnesium ~chemistNATE (2:54) [here]
Ions
Mass Spectrometry
Mass Spectrometry|Atomic Structure & Properties ~Kahn Academy (4:17) [here]
Groups (aka Families) of Elements
Physical & Chemical Changes
Coulomb's Law
Bond Length & Bond Energy
Naming Compounds
Multivalent Ionic Compounds
Ionic Compounds
Hydrates
Naming Acids
Naming Organic Compounds
|
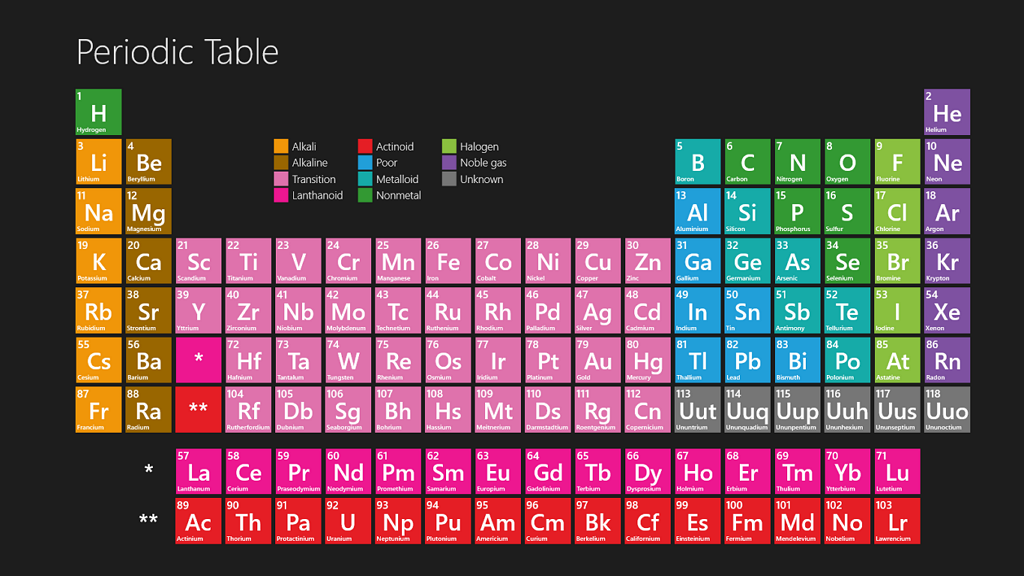
Practice Periodic Table
Naming Compounds
Formula Writing Guided Practice
Naming Organic & Biological Molecules
Molecule vs Formula Unit
Answers to Selected Exercises
|
Resources Resources for Class
What do I need to Know for this Unit?
Periodic Table
Active Learning
By the end of the course you will be able to name and write chemical formulas at the introductory college level.
Naming Compounds
Ion Resources
Acids
|
|
History of the Atom Scientists
|
Memorize these 12 Polyatomic Ions for the Quiz:
Ionic Formulas and Naming Flashcards:
Bonding |
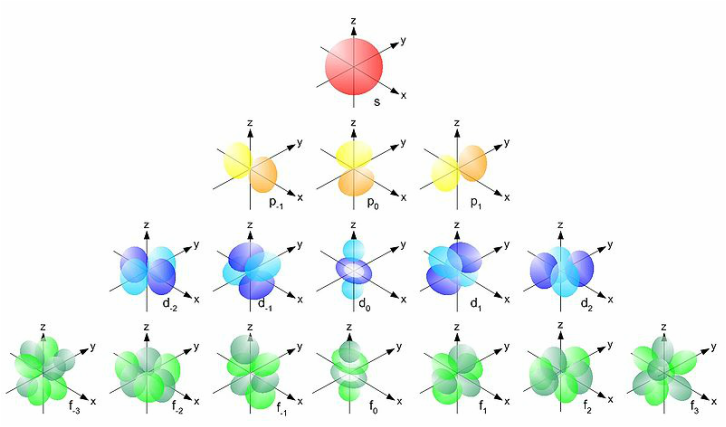
Atomic Orbitals:
|
Lab - Unknown Lab Information
|

Classes of Organic Compounds Study Set
Elements and Abundance
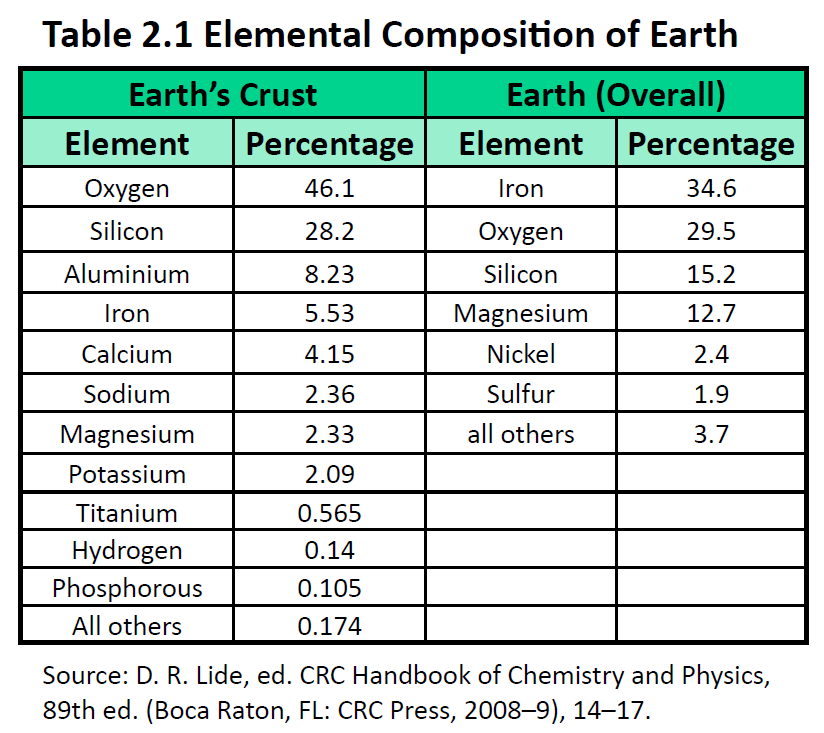
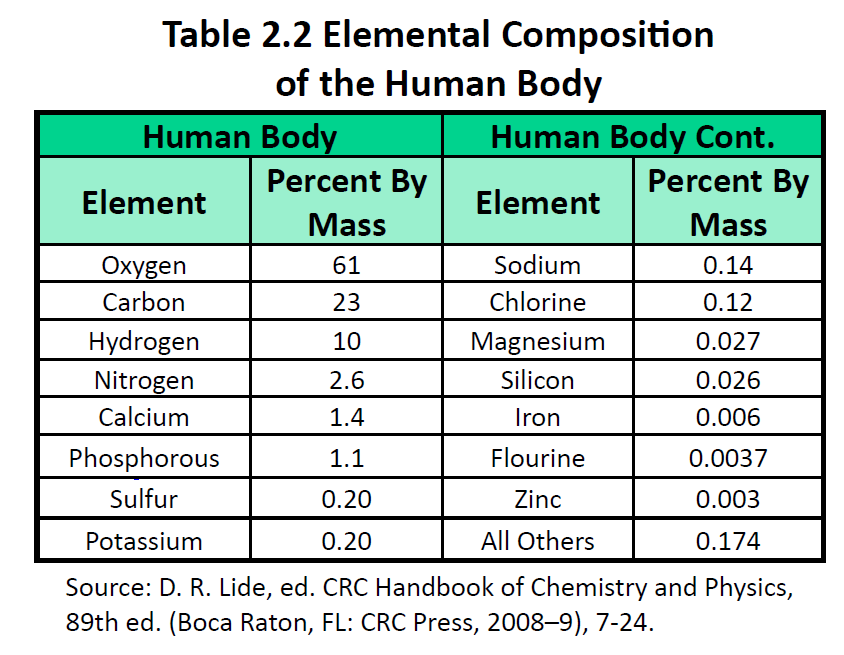
The elements vary widely in abundance. In the universe as a whole, the most common element is hydrogen (about 90%), followed by helium (most of the remaining 10%). All other elements are present in relatively minuscule amounts, as far as we can detect. On the planet Earth, however, the situation is rather different. Oxygen makes up 46.1% of the mass of Earth’s crust (the relatively thin layer of rock forming Earth’s surface), mostly in combination with other elements, while silicon makes up 28.5%. Hydrogen, the most abundant element in the universe, makes up only 0.14% of Earth’s crust. Table 2.1 “Elemental Composition of Earth” lists the relative abundances of elements on Earth as a whole and in Earth’s crust. Table 2.2 “Elemental Composition of a Human Body” lists the relative abundances of elements in the human body. If you compare Table 2.1 “Elemental Composition of Earth” and Table 2.2 “Elemental Composition of a Human Body”, you will find disparities between the percentage of each element in the human body and on Earth. Oxygen has the highest percentage in both cases, but carbon, the element with the second highest percentage in the body, is relatively rare on Earth and does not even appear as a separate entry in Table 2.1 “Elemental Composition of Earth”; carbon is part of the 0.174% representing “other” elements. How does the human body concentrate so many apparently rare elements?
The relative amounts of elements in the body have less to do with their abundances on Earth than with their availability in a form we can assimilate. We obtain oxygen from the air we breathe and the water we drink. We also obtain hydrogen from water. On the other hand, although carbon is present in the atmosphere as carbon dioxide, and about 80% of the atmosphere is nitrogen, we obtain those two elements from the food we eat, not the air we breathe
Source: Western Oregon University
The relative amounts of elements in the body have less to do with their abundances on Earth than with their availability in a form we can assimilate. We obtain oxygen from the air we breathe and the water we drink. We also obtain hydrogen from water. On the other hand, although carbon is present in the atmosphere as carbon dioxide, and about 80% of the atmosphere is nitrogen, we obtain those two elements from the food we eat, not the air we breathe
Source: Western Oregon University