Unit 9 Electrochemistry
Electrochemistry is a science that deals with the relation of electricity to chemical changes and with the interconversion of chemical and electrical energy.
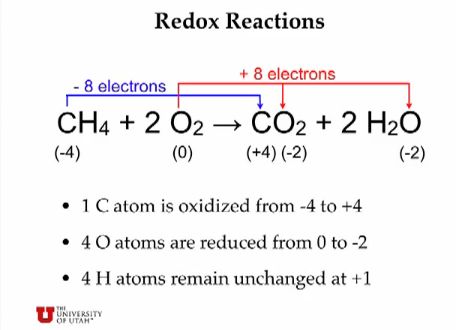
Oxidation is defined as the interaction between oxygen molecules and all the different substances they may contact, from metal to living tissue. Technically, however, with the discovery of electrons, oxidation came to be more precisely defined as the loss of at least one electron when two or more substances interact. Those substances may or may not include oxygen. (Incidentally, the opposite of oxidation is reduction — the addition of at least one electron when substances come into contact with each other.) ~wiseGEEK
Big Idea 3 - Changes in matter involve the rearrangement and/or reorganization of atoms and/or the transfer of electrons.
Oxidation is defined as the interaction between oxygen molecules and all the different substances they may contact, from metal to living tissue. Technically, however, with the discovery of electrons, oxidation came to be more precisely defined as the loss of at least one electron when two or more substances interact. Those substances may or may not include oxygen. (Incidentally, the opposite of oxidation is reduction — the addition of at least one electron when substances come into contact with each other.) ~wiseGEEK
Big Idea 3 - Changes in matter involve the rearrangement and/or reorganization of atoms and/or the transfer of electrons.
Topics Covered:
- Electrochemical cells and voltage
- Standard Conditions
- Constructing voltaic cells
- Relating to thermo and equilibrium
- Nernst Equation (qualitative and quantitative)
- Electrolysis
- Applications of electrochemical cells
The Oxygen Irony. Oxygen is not discriminating about what other molecular structures it might destroy in its quest to become electrically whole. If there are electrons to spare in a protein molecule, or in the fats that make up a cells’ membrane, or even in the DNA that is crucial to the functioning of our cells and our bodies,...
Oxygen. Critical to life, but can be very destructive... The Oxygen Irony
Oxygen. Critical to life, but can be very destructive... The Oxygen Irony
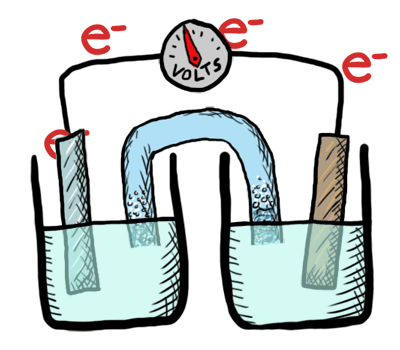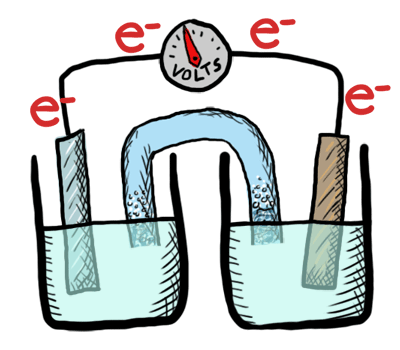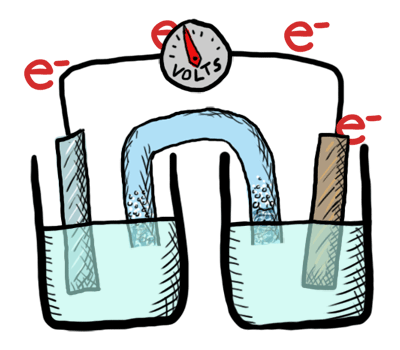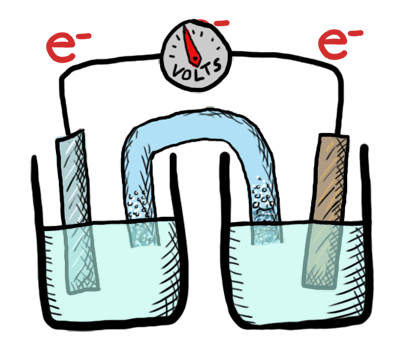
Electrochemistry involves TWO MAIN TYPES of electrochemical cells:
- Galvanic (voltaic) cells: (+) E cell - which are thermodynamically favorable chemical reactions (“battery").
- Electrolytic cells: (-) E cell - which are thermodynamically UNfavorable and require external e- source (a direct current or DC power source). (“needs a battery”)
|
|
|
Videos
RedOx Reactions
Balancing Redox Equations
Introduction to Electrochemistry
Cell Potentials
AP Chemistry Electrochemistry: Cell Potentials (10:42) Shows how to calculate galvanic (voltaic) cell potentials (voltages) using Standard Reduction Potentials.
Energy, Cell Potentials and Gibbs Free Energy Relationships
AP Chemistry Electrochemistry - Relating E, G, and K (9:28)
Relating delta G with the E (potential) of a voltaic cell, and relating the E of the cell to the equilibrium constant (K). Galvanic (voltaic) Cells |
Tutorials
Oxidation
Practice
Oxidation Reduction aka RedOx
Net Ionic Equations
Electrochemistry
More Videos:
Chemistry Music Video #27
You Start at the Anode (3:28) This song describes the workings of a voltaic cell, tracing the path of negative charge (external circuit: electrons through the wire, internal circuit: anions through the salt bridge). [here] |
Classroom Resources
Dr. Leggett Electrochemistry Videos
|
|
Galvanic (Voltaic) Cell Animation:
Electrolytic CellsElectroplating
Electrolysis
|
Simulations
|
Tutorial Notes
|
Balancing Redox Reactions (Step-by-Step)
Balancing redox reactions is slightly more complex than balancing standard reactions, but still follows a relatively simple set of rules.
One major difference is the necessity to know the half-reactions of the involved reactants; a half-reaction table is very useful for this. Half-reactions are often useful in that two half reactions can be added to get a total net equation. Although the half-reactions must be known to complete a redox reaction, it is often possible to figure them out without having to use a half-reaction table. This is demonstrated in the acidic and basic solution examples.
Besides the general rules for neutral conditions, additional rules must be applied for aqueous reactions in acidic or basic conditions.
The method used to balance redox reactions is called the Half Equation Method. In this method, the equation is separated into two half-equations; one for oxidation and one for reduction.
Each equation is balanced by adjusting coefficients and adding H2O, H+, and e- in this order:
- Balance elements in the equation other than O and H.
- Balance the oxygen atoms by adding the appropriate number of water (H2O) molecules to the opposite side of the equation.
- Balance the hydrogen atoms (including those added in step 2 to balance the oxygen atom) by adding H+ ions to the opposite side of the equation.
- Add up the charges on each side. Make them equal by adding enough electrons (e-) to the more positive side. (Rule of thumb: e- and H+ are almost always on the same side.)
- The e- on each side must be made equal; if they are not equal, they must be multiplied by appropriate integers (the lowest common multiple) to be made the same.
- The half-equations are added together, canceling out the electrons to form one balanced equation. Common terms should also be canceled out.
- (If the equation is being balanced in a basic solution, through the addition of one more step, the appropriate number of OH- must be added to turn the remaining H+into water molecules.)
- The equation can now be checked to make sure that it is balanced
These steps as well as example problems can be found at LibreText Balancing Redox Reactions
Oxidation-Reduction Reactions: Balancing
Review: Balancing chemical equations; oxidation-reduction.
Learning Goals:
Review: Balancing chemical equations; oxidation-reduction.
Learning Goals:
- Identify the oxidizing agent (oxidant) and reducing agent (reductant) in an oxidation-reduction reaction.
- Balance oxidation-reduction reactions by the method of half-reactions.
Voltaic Cells: Standard Electrode and Cell Potentials
Review: Electrolyte solutions.
Learning Goals:
Review: Electrolyte solutions.
Learning Goals:
- Diagram simple voltaic and electrolytic cells, labeling the anode, the cathode, the directions of ion and electron movement, and the signs of the electrodes.
- Given appropriate standard reduction potentials, calculate the cell potential generated by voltaic cell at standard state conditions.
- Use standard reduction potentials to predict whether a reaction will be spontaneous at standard state conditions.
Relationship of EMF to Concentration, Free-Energy Changes, and the Equilibrium Constant
Review: Equilibrium Constant; free-energy function; free energy and the equilibrium state.
Review: Equilibrium Constant; free-energy function; free energy and the equilibrium state.
Practical Examples of Electrochemistry
Learning Goals:
Learning Goals:
- Describe the lead-acid storage battery, and the alkaline, nickel-cadmium, and lithium ion batteries.
- Descrive corrosion in terms of the electrochemistry involved, and explain the principles that underlie cathodic protection.
Electrolysis: Faraday's Law
Learning Goals:
Learning Goals:
- Diagram simple electrolytic cells, labeling the anode, the cathode, the directions of ion and electron movement, and the sign of the electrodes.
- Given appropriate electrode potentials, calculate the minimum emf required to caus electrolysis.
- Relate time, current, and the amount of substance produced or consumed in an electrolysis reaction and given two of the three quantities, you should be able to calculate the third.
- Calculate the maximum electrical work performed by a voltaic cell, and the minimum electrical work required for an electrolytic process.