Unit 7: Quantum Theory, Electron Configuration, & Periodicity
|
Videos
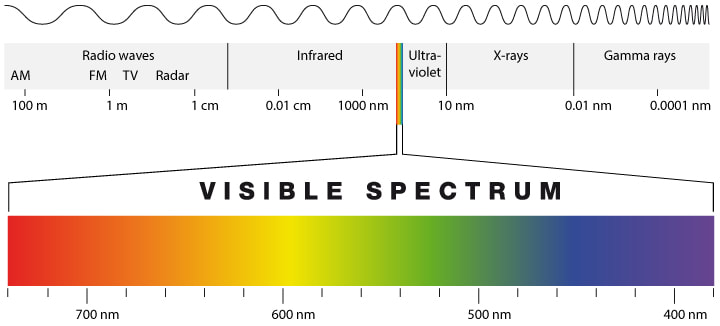
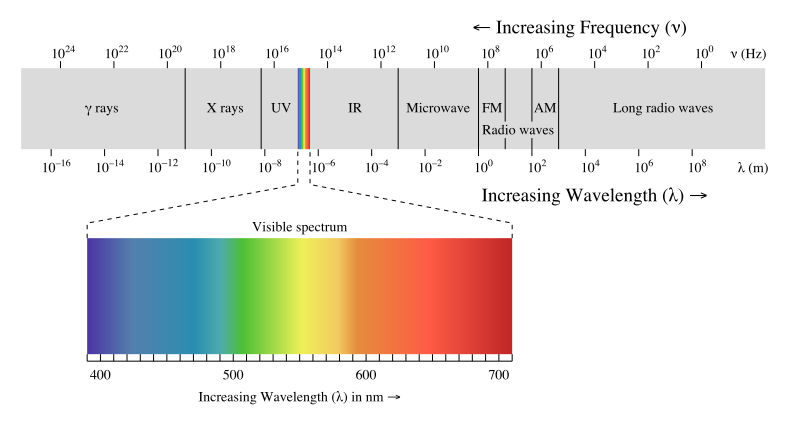
Electromagnetic Radiation
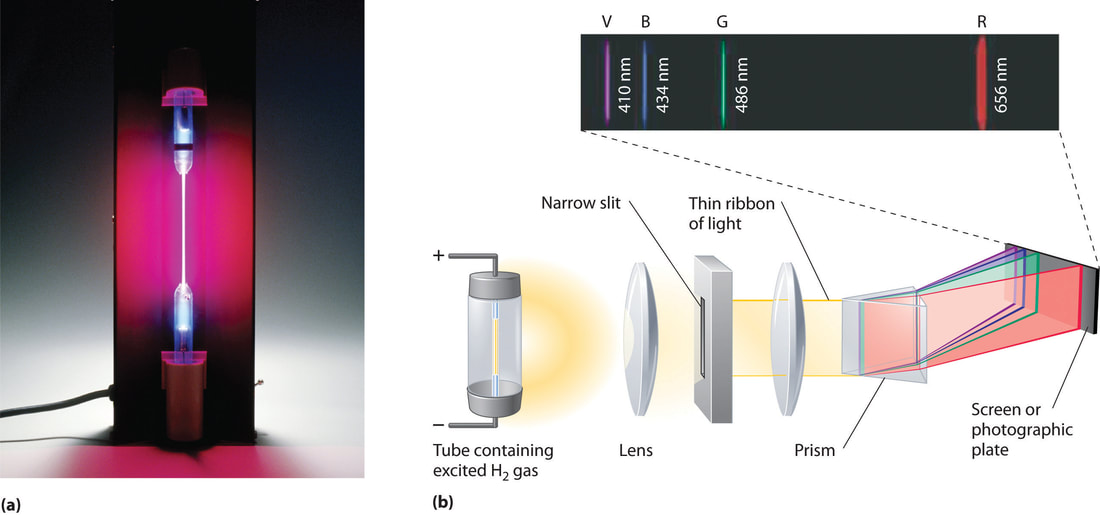
Photo Electron Spectroscopy (PES)
& Coulomb’s Law Photoelectron Spectroscopy (PES) for AP Chem Review ~Brad Palmer (12:52) [more here]
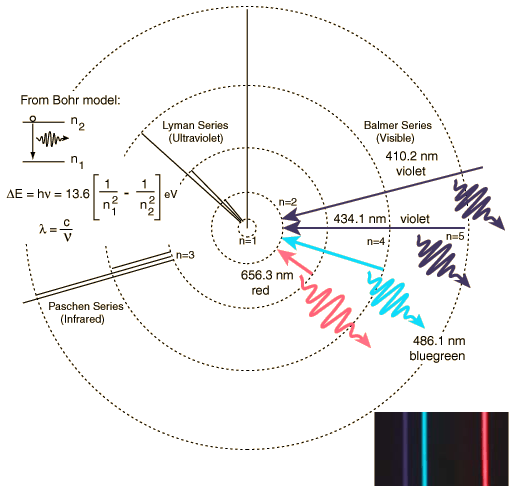
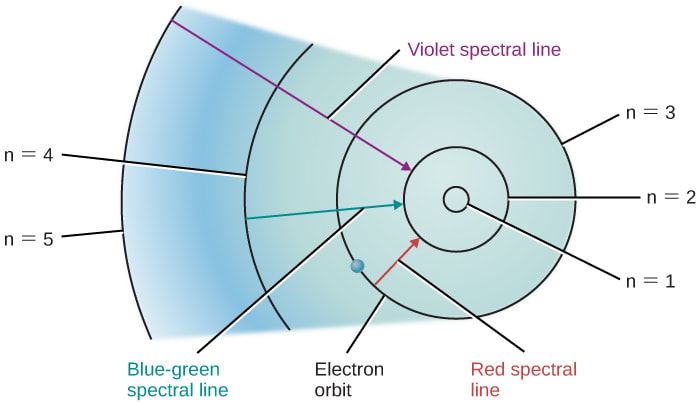
Quantum Mechanical Model - Bozeman Science #007 (4:35) How the quantum mechanical model of the atom refined the shell model. Uncertainty of the position of the electron as well as spin forces create an improved model where the position of the electron is determined through quantum numbers... [more here]
Schrodinger’s Cat Explained - London City Girl Knowledge (4:01)... [more here]
Orbitals
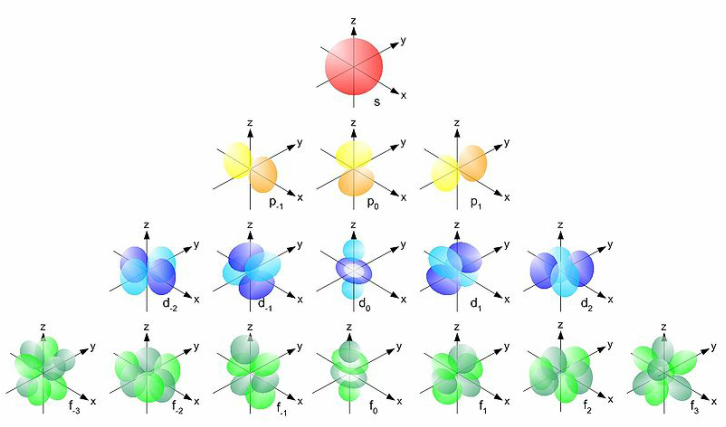
Orbitals - Crash Course Chemistry #25 (10:51) Hank discusses what Molecules actually look like and why, some quantum-mechanical three dimensional wave functions are explored, he touches on hybridization, and delves into sigma and pi bonds. [more here...]
Valence Electrons
Valence Electrons & the Periodic Table (16:52) Where do electrons live in atoms? They live in energy levels or shells, which are varying distances from the nucleus, and can hold varying numbers of electrons. The most important electrons in an atom are the valence electrons, which are in the outermost energy level or shell. We'll look at how to determine the number of valence electrons in an atom, based on which column it is in on the periodic table...[’more here]
Electron Configuration
Electron Configuration - Bozeman Science #005 (10:16) How to write out the electron configuration for atoms on the period table. & Why electrons arrange themselves in shells, subshells and orbitals by using Coulomb's law and studying the first ionization energies of different atoms... [more here]
Uncertainty
The Uncertain Location of Electrons ~Ted-Ed (3:46) The tiny atoms that make up our world are made up of even tinier protons, neutrons and electrons. Though the number of protons determine an atom's identity, it's the electrons -- specifically, their exact location outside the nucleus -- that particularly perplex scientists. George Zaidan and Charles Morton show how to make an educated guess of where those itty-bitty freewheeling electrons might be. [Link Here]
Paramagnetic and Diamagnetic
Liquid Nitrogen vs. Liquid Oxygen: Magnetism ~Jefferson Lab (2:49)
What happens when liquid nitrogen and liquid oxygen are exposed to a strong magnetic field? [Link Here] |
Tutorials
Electron Configuration
LebreTexts on Electron Configuration
The electron configuration of an atomic species (neutral or ionic) allows us to understand the shape and energy of its electrons. Many general rules are taken into consideration when assigning the "location" of the electron to its prospective energy state, . Knowing the electron configuration of a species gives us a better understanding of its bonding ability, magnetism and other chemical properties. [more here] Step-by-Step of How to Write Electron Configurations for atoms of any element WikiHow [more here]
Simulations
Build an Atom - PhET - atoms, atomic structure and isotope symbols... [more here]
Periodic Trends
Periodic Trends: Ionization Energy, Atomic Radius, Ionic Radius ~AACT Chemistry Solutions
In this simulation, students can investigate the periodic trends of atomic radius, ionization energy, and ionic radius. By choosing elements from the periodic table, atoms can be selected for a side by side comparison and analysis. Students can also attempt to ionize an atom by removing its valence electrons. Quantitative data is available for each periodic trend, and can be further examined in a graph. Photoelectron Spectroscopy
Periodicity
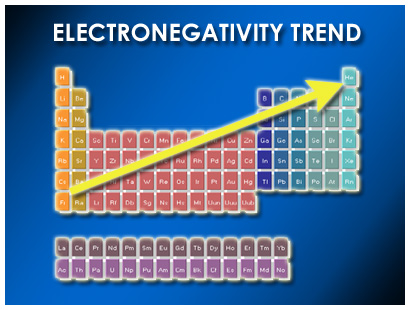
Periodicity - Bozeman Science #006 (8:56) Why atoms in the periodic table show trends in ionization energy, atomic radii, electronegativity and charge. All of these trends are explained through Coulomb's Law... [more here]
The Periodic Table - Atomic Radius, Ionization Energy, and Electronegativity ~Professor Dave Explains (7:52)... [more here]
Coulomb’s Law - Bozeman Science #004 (10:57) How we can use Coulomb's law to predict the structure of atoms and measure Ionization Energy through the use of Photoelectron Spectroscopy (PES). Electron's are held around the nucleus because of differences in charge. As the atoms become larger the charges change and so do the structures... [more here]
|
Class Information
Order of Fill:
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, 8s Answers
Photoelectric Effect
Photoelectric Effect Demonstration
The Photoelectric effect helps to illustrate the wave-particle duality of Light. ~National STEM Centre (3:04) [Link Here] Wave-Particle Duality
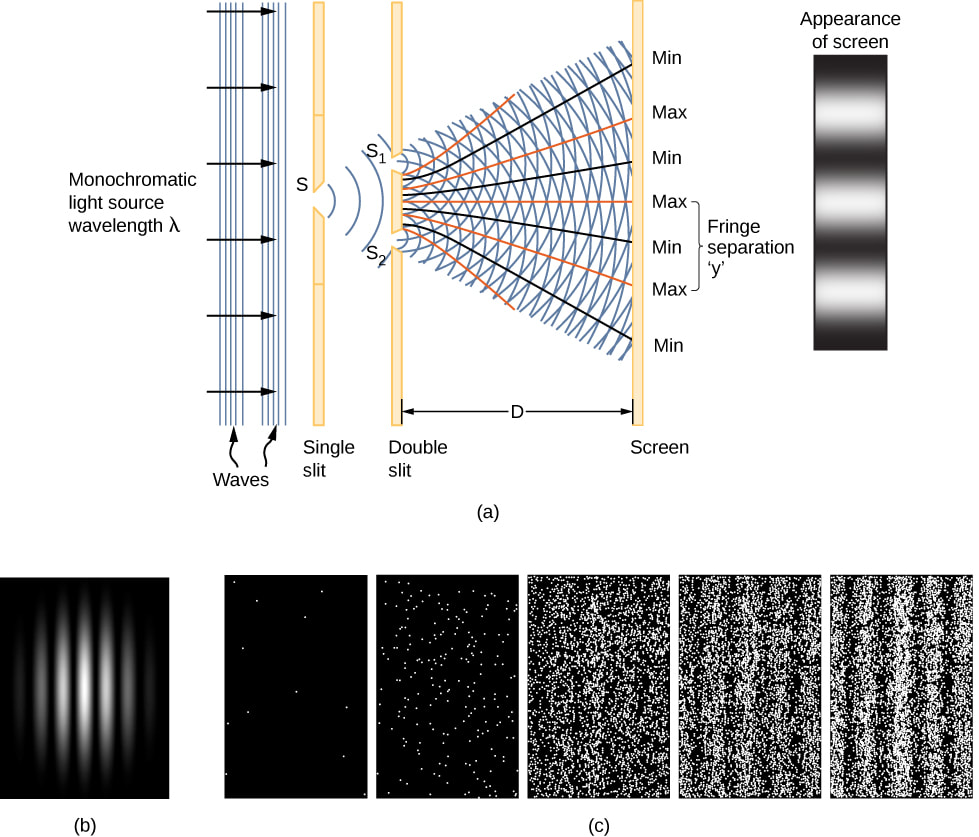
Wave-Particle Duality Part 1 ~Bozeman (6:09) Paul Andersen explains the wave-particle duality discovered by scientists. In certain situations particles (like electrons and photons) display wave like properties. This phenomenon can best be explored using the double slit experiment. Small particles follow the rules of quantum mechanics creating results that are not very intuitive. [Link Here]
Wave-Particle Duality Part 2 ~Bozeman (4:44) Paul Andersen explains how classical waves (like light) can have particle properties. Albert Einsetein used the photoelectric effect to show how photons have particle properties. [Link Here]
Schrödinger's cat: A thought experiment in quantum mechanics - Chad Orzel (4:37) Austrian physicist Erwin Schrödinger, one of the founders of quantum mechanics, posed this famous question: If you put a cat in a sealed box with a device that has a 50% chance of killing the cat in the next hour, what will be the state of the cat when that time is up? Chad Orzel investigates this thought experiment. [video found here]
[full lesson here] |