Unit 5 - Major Classes of Reactions in Aqueous Solutions
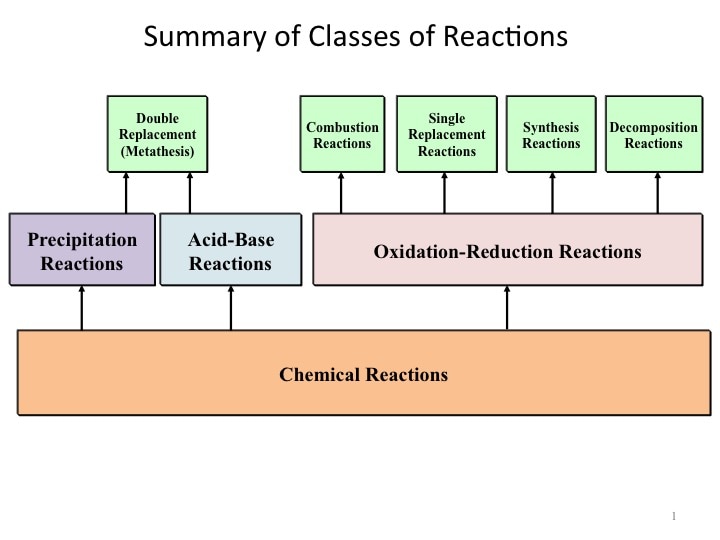
In this unit we are going to focus on the 3 Classes of chemical reactions; Precipitation Reactions, Acid-Base Reactions, and Oxidation-Reduction Reactions (Redox).
The following list is a GUIDE to what you should study in order to be prepared for the test on TOPIC 5
In order to be fully prepared you should seek help if required, refer to the relevant slides in the and review ALL relevant notes, homework, worksheets, classwork and other materials.
For this unit, ALL students should;
In order to be fully prepared you should seek help if required, refer to the relevant slides in the and review ALL relevant notes, homework, worksheets, classwork and other materials.
For this unit, ALL students should;
- Understand that a reaction in aqueous solution is one that is carried out in water
- Understand the terms electrolyte, weak electrolyte and non-electrolyte
- Understand the difference between, and be able to write, full, ionic and net ionic equations
- Learn and be able to apply solubility rules
- Recall that an acid can be defined as a hydrogen ion donor
- Recall that a base can be defined as a hydrogen ion acceptor
- Understand how the degree of ionization (dissociation) determines the strength of an acid and a base
- Understand that in a neutralization reaction an acid and base react to form a salt and water
- Understand that oxidation and reduction can be described in terms of loss and gain of electrons respectively
- Understand and be able to apply the Oxidation Number concept
- Understand the concept of disproportionation
- Understand and be able to recognize the different types of REDOX reaction. Namely synthesis (combination), decomposition, combustion, single and double displacement (replacement) including metal displacement, hydrogen displacement from water and acids and halogen displacement
- Learn and be able to use the reactivity series as a tool for predicting displacement reactions
- Be able to perform REDOX titration calculations
|
Videos
Types of Chemical Reactions
Every second, billions of reactions are happening around you. Every single reaction can be placed into 1 of 5 categories. Here we help you learn the basics of a vast world you can’t see.
Indications of a Chemical Reaction
|
Practice/Worksheets/Games
Precipitation Reactions
Redox Reactions
|
Other Resources
From Our Classroom:
Labs
Solubility
|
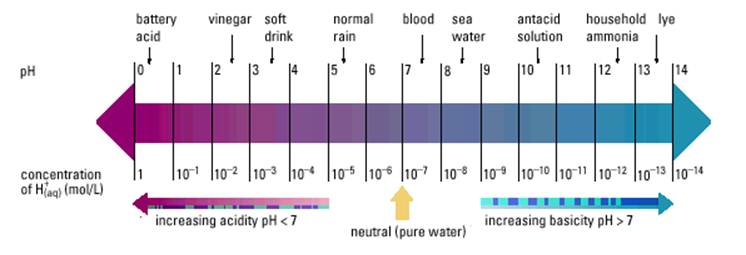
As you can see from the above diagram, an increase of one in pH is a tenfold increase in [OH].
A solution with a pH of 4 is 10 times more acidic than a solution with a pH of 5.
A solution with a pH of 3 is 100 times more acidic than a solution with a pH of 5.
A solution with a pH of 2 is 1000 times more acidic than a solution with a pH of 5.
A solution with a pH of 4 is 10 times more acidic than a solution with a pH of 5.
A solution with a pH of 3 is 100 times more acidic than a solution with a pH of 5.
A solution with a pH of 2 is 1000 times more acidic than a solution with a pH of 5.
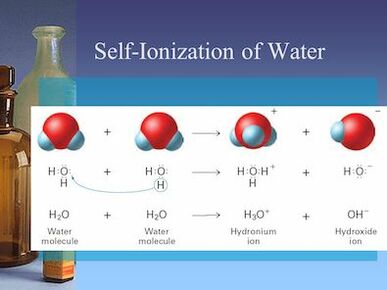
Ionization of Water
|
Videos
Reactions
How It's Made - Uranium (5:21) [here]
Double Replacement: Precipitation
Double Replacement: Acid/Base
pH - pOH
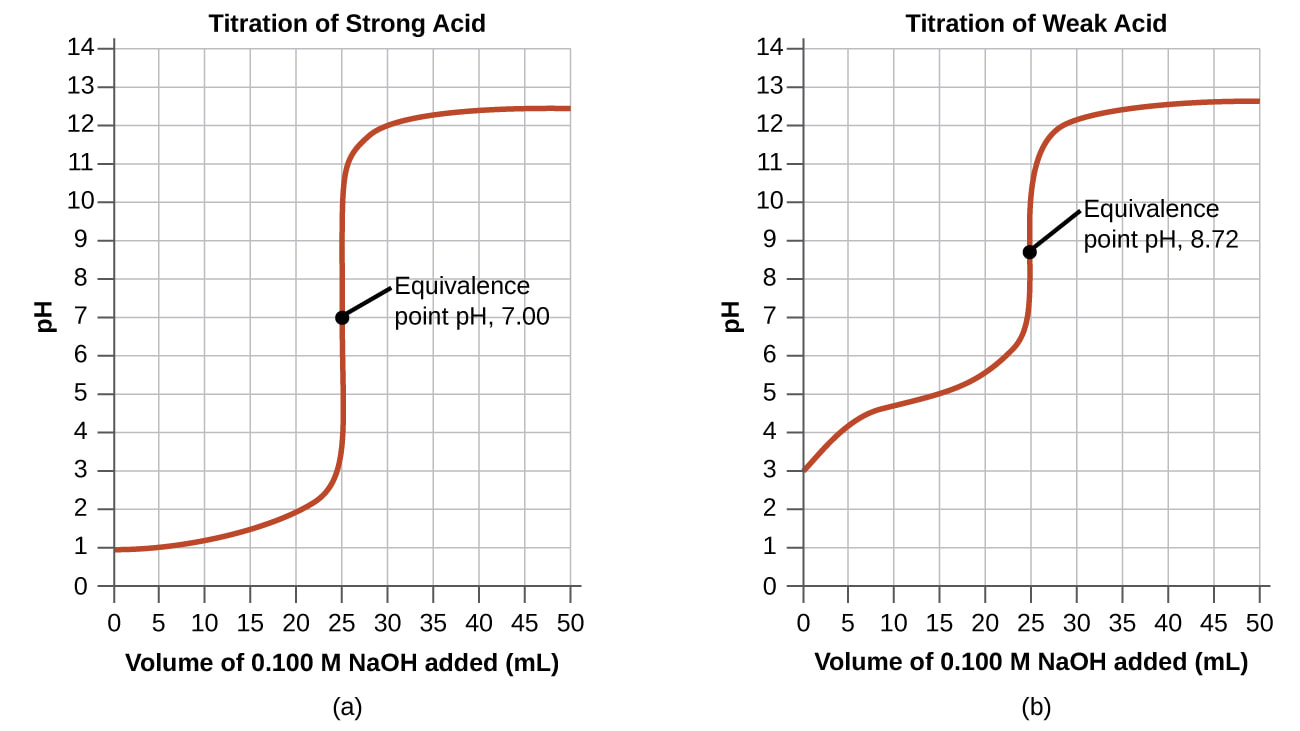
Titrations
Redox Reactions
Introduction to Oxidation Reduction (Redox) Reactions - Tyler DeWitt (13:04) This is an introduction to oxidation reduction reactions, which are often called redox reactions for short. An oxidation reduction (redox) reaction happens when electrons are transferred between atoms... [more info]
The Oxidation Reduction Question That Tricks Everyone - Tyler DeWitt (6:18) Don't make the most common mistake in Oxidation Reduction! This question confuses so many students. Watch this video to learn how to do this right. In chemistry, reduction is the gain of electrons, and oxidation is the loss...
[more info] Oxidizing Agents & Reducing Agents - Tyler DeWitt (14:55) Learn about oxidizing agents and reducing agents, what they are, what they do, and how to identify them in chemical equations... [more info]
How to Calculate Oxidation Numbers Introduction - Tyler DeWitt (13:25) Learn how to determine the oxidation numbers or oxidation states for the elements in a chemical compound. The oxidation numbers tell us how electrons are divided up or shared between atoms in a chemical compound. The oxidation numbers also tell us how electrons move in an oxidation reduction (redox) reaction. There are a set a rules that we use to determine oxidation number... [more info]
How to Calculate Oxidation Number Practice Problems - Tyler DeWitt (15:24) Practice problems for how to calculate and determine oxidation numbers, (oxidation states) For neutral compounds, the oxidation numbers add up to zero. For polyatomic ions, the oxidation numbers add up to charge of the ion. [more info]
How to Find the Oxidation Number for C in C2H2 (ethyne) ~Wayne Breslyn (1:07)
"Dr. B" uses a few rules and simple math, and a ball and stick model to explain how to find the correct oxidations numbers for each element in the molecule. [more info] |
Simulations
Acid-Base Titration Simulations
Learning by Simulations: Acid-Base Titration [here]
pH
Acid-Base Reactions
Titrations
Oxidation Numbers
Oxidation/Reduction (Redox) Reactions
Half Reaction Method
Answers to Class Practice
WS 5.4 Predicting Precipitation KEY
WS 5.6 Acids/Bases KEY
WS 5.7 Acid Base Titrations
WS 5.10 Writing Half Reactions KEY
Pipelines in the US
How to Balance Redox Equations in Acidic Solution - Tyler DeWitt (14:59) Step by step how to balance an oxidation reduction (redox) reaction in acidic solution. Most important - both charges and atoms must balance... [more info]
How to Balance Redox Equations in Acidic Solution Example 1 - Tyler DeWitt (14:45) Step by step through how to balance an oxidation reduction (redox) reaction in acidic solution... [more info]
Balance Redox Equations in Acid Example 2 (Advanced) - Tyler DeWitt (15:54) How to balance an oxidation reduction (redox) reaction in acidic solution. This example is advanced.... [more info]
How to Balance Redox Reactions in Basic Solution - Tyler DeWitt (17:59) How to balance an oxidation reduction (redox) reaction in basic solution. The process is similar to balance an oxidation reduction equation in acidic solution. [more info]
|
Rules for Assigning Oxidation Numbers
- The convention is that the cation is written first in a formula, followed by the anion. For example, in NaH, the H is H-; in HCl, the H is H+.
- The oxidation number of a free element is always 0. The atoms in He and N2, for example, have oxidation numbers of 0.
- The oxidation number of a monatomic ion equals the charge of the ion. For example, the oxidation number of Na+ is +1; the oxidation number of N3- is -3.
- The usual oxidation number of hydrogen is +1. The oxidation number of hydrogen is -1 in compounds containing elements that are less electronegativethan hydrogen, as in CaH2.
- The oxidation number of oxygen in compounds is usually -2. Exceptions include OF2 because F is more electronegative than O, and BaO2, due to the structure of the peroxide ion, which is [O-O]2-.
- The oxidation number of a Group IA element in a compound is +1.
- The oxidation number of a Group IIA element in a compound is +2.
- The oxidation number of a Group VIIA element in a compound is -1, except when that element is combined with one having a higher electronegativity. The oxidation number of Cl is -1 in HCl, but the oxidation number of Cl is +1 in HOCl.
Helmenstine, Anne Marie, Ph.D. "What Are the Rules for Assigning Oxidation Numbers?" ThoughtCo, Aug. 28, 2020, thoughtco.com/rules-for-assigning-oxidation-numbers-607567.
Oxidation Reduction Reactions—Redox Reactions
This is an introduction to oxidation-reduction reactions, also known as redox reactions. Learn what redox reactions are, get examples of oxidation-reduction reactions, and find out why redox reactions are important.
What Is an Oxidation-Reduction or Redox Reaction? Any chemical reaction in which the oxidation numbers (oxidation states) of the atoms are changed is an oxidation-reduction reaction. Such reactions are also known as redox reactions, which is shorthand for reduction-oxidation reactions.
Oxidation and Reduction Oxidation involves an increase in oxidation number, while reduction involves a decrease in oxidation number. Usually, the change in oxidation number is associated with a gain or loss of electrons, but there are some redox reactions (e.g., covalent bonding) that do not involve electron transfer. Depending on the chemical reaction, oxidation and reduction may involve any of the following for a given atom, ion, or molecule:
Example of an Oxidation-Reduction Reaction The reaction between hydrogen and fluorine is an example of an oxidation-reduction reaction:
H2 + F2 → 2 HF
The overall reaction may be written as two half-reactions:
H2 → 2 H+ + 2 e− (the oxidation reaction)
F2 + 2 e− → 2 F− (the reduction reaction)
There is no net change in charge in a redox reaction so the excess electrons in the oxidation reaction must equal the number of electrons consumed by the reduction reaction. The ions combine to form hydrogen fluoride:
H2 + F2 → 2 H+ + 2 F− → 2 HF
Importance of Redox Reactions The electron transfer system in cells and oxidation of glucose in the human body are examples of redox reactions. Oxidation-reduction reactions are vital for biochemical reactions and industrial processes as well. Redox reactions are used to reduce ores to obtain metals, to produce electrochemical cells, to convert ammonia into nitric acid for fertilizers, and to coat compact discs.
ThoughtCo
Helmenstine, Anne Marie, Ph.D. "Oxidation Reduction Reactions—Redox Reactions." ThoughtCo, Feb. 16, 2021, thoughtco.com/oxidation-reduction-reactions-604037.
What Is an Oxidation-Reduction or Redox Reaction? Any chemical reaction in which the oxidation numbers (oxidation states) of the atoms are changed is an oxidation-reduction reaction. Such reactions are also known as redox reactions, which is shorthand for reduction-oxidation reactions.
Oxidation and Reduction Oxidation involves an increase in oxidation number, while reduction involves a decrease in oxidation number. Usually, the change in oxidation number is associated with a gain or loss of electrons, but there are some redox reactions (e.g., covalent bonding) that do not involve electron transfer. Depending on the chemical reaction, oxidation and reduction may involve any of the following for a given atom, ion, or molecule:
- Oxidation involves the loss of electrons or hydrogen OR gain of oxygen OR increase in oxidation state.
- Reduction involves the gain of electrons or hydrogen OR loss of oxygen OR decrease in oxidation state.
Example of an Oxidation-Reduction Reaction The reaction between hydrogen and fluorine is an example of an oxidation-reduction reaction:
H2 + F2 → 2 HF
The overall reaction may be written as two half-reactions:
H2 → 2 H+ + 2 e− (the oxidation reaction)
F2 + 2 e− → 2 F− (the reduction reaction)
There is no net change in charge in a redox reaction so the excess electrons in the oxidation reaction must equal the number of electrons consumed by the reduction reaction. The ions combine to form hydrogen fluoride:
H2 + F2 → 2 H+ + 2 F− → 2 HF
Importance of Redox Reactions The electron transfer system in cells and oxidation of glucose in the human body are examples of redox reactions. Oxidation-reduction reactions are vital for biochemical reactions and industrial processes as well. Redox reactions are used to reduce ores to obtain metals, to produce electrochemical cells, to convert ammonia into nitric acid for fertilizers, and to coat compact discs.
ThoughtCo
Helmenstine, Anne Marie, Ph.D. "Oxidation Reduction Reactions—Redox Reactions." ThoughtCo, Feb. 16, 2021, thoughtco.com/oxidation-reduction-reactions-604037.