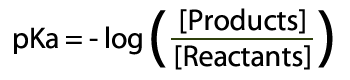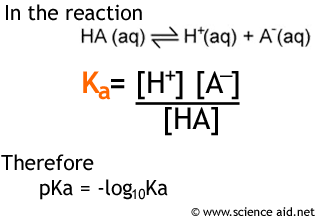Unit 14 Acid Base Equilibria
"Your blood is a buffered solution. If it wasn't, your pH would go way down every time you had a soda and way up whenever you took some Tums. It's not a very handy survival technique to die every time you have a soda." ~ Mr. Guch
|
Videos
Bozeman AP Chemistry Videos in Order:
Reversible Reactions
062 - Reversible Rxns How reversible reactions achieve equilibrium as reactants are converted to products and products are converted to reactants. A model shows how forward reaction rates and reverse reactions rates approach equality at equilibrium. Physical, chemical, biological, and ecological examples of reversible reactions are included. The Reaction Quotient
063 - The Rxn Quotient How the reaction quotient is used to determine the progress of a reversible reaction. The reaction quotient (Q) is the ratio of the concentration of products to the concentration of reactants. The reaction quotient will equal the equilibrium constant when the reaction is at equilibrium. (7:08) |
Practice Tests
K - The Equilibrium Constant
This page is an exercise in calculating the equilibrium constant. K the Equilibrium Constant This page is an exercise in calculating an equilibrium concentration from a balanced equation, the K value and a partial set of equilibrium concentration.
Calculating an Equilibrium Concentration Tutorials
Calculations of pH, pOH, [H+] & [OH-]
Great Practice Tutorial by ScienceGeek - You will need a calculator. Practice Worksheets
Henderson Hasselbalch Equation
Very Useful Videos
|
Other Resources
From Mrs. Forest’s Classroom:
Student Guides
Acid-Base Equilibria
Additional Aspects of Acid-Base Equilibria
Simulations
Extra Help
|
|
pH Curve Calculator
Calculate the pH during titration of a mixture and trace the pH curve. |