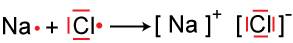Unit 8: Bonding & Molecular Geometry
The Chemical Bond
Atoms combine to form compounds. In general, the study of chemical bonds involves the consideration of the forces that link atoms together. In a broad sense, bonding can be considered to be either ionic or covalent, representing two extremes in a continuity of bond types. A theory of bonding should help to rationalize the shapes of molecules as well as their stoichiometries.
Atoms combine to form compounds. In general, the study of chemical bonds involves the consideration of the forces that link atoms together. In a broad sense, bonding can be considered to be either ionic or covalent, representing two extremes in a continuity of bond types. A theory of bonding should help to rationalize the shapes of molecules as well as their stoichiometries.
|
Videos Drawing Lewis Dot Structures
How to Draw Lewis Structures:
Five Easy Steps ~Wayne Breslyn Video tutorial for how to draw Lewis Structures. Covers the basic Lewis structures you'll see in an introductory chemistry class. (5:56) [here] Valence Electrons & Lewis Dot Structures
Dr. B’s Chemistry Videos
Look around, you’ll find a lot of videos on Lewis Dot Structures you can use. Very easy to understand! Dr. B’s Chemistry Videos on YouTube Lewis Electron Dot Diagram - BrightStorm (9:22)... [more info]
Drawing Lewis Dot Diagrams - Bozeman (9:41)... [more info]
Bonds
Chemistry Music Video 16: What Kinds of Bonds are Those? This song describes ionic bonds and covalent bonds, and describes the difference between polar and nonpolar covalent bonds...
[more info] |
Practice Lewis Structure Multiple Choice Exercise 20 Questions to test your Lewis Structure prowess! by Science Geek [more info]
Lewis Structures
Writing Lewis Structures by Trial and Error. Includes Step-by-Step Instructions (Purdue Walton) ... [more info] Building Molecules with Lewis Dot Structures - Try building these structures then click on the molecule to reveal the answer... [more info]
|
Other Resources Class
NASL Method for Writing Lewis Structures [here]
Tutorials
Simulations
|
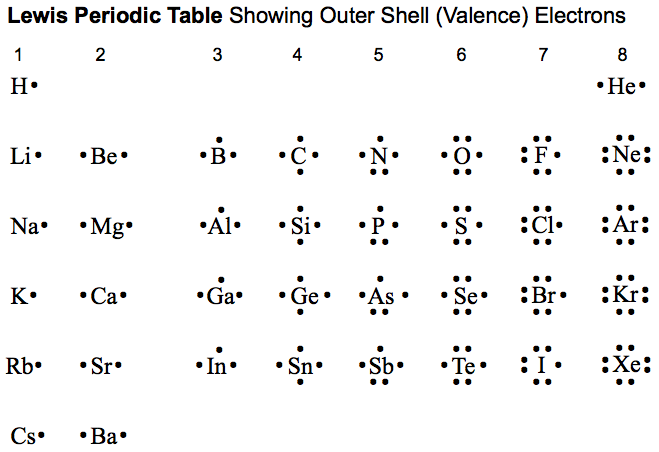
Lewis Diagrams of the Elements
Notice that each element has the same # of valence electrons as their group #
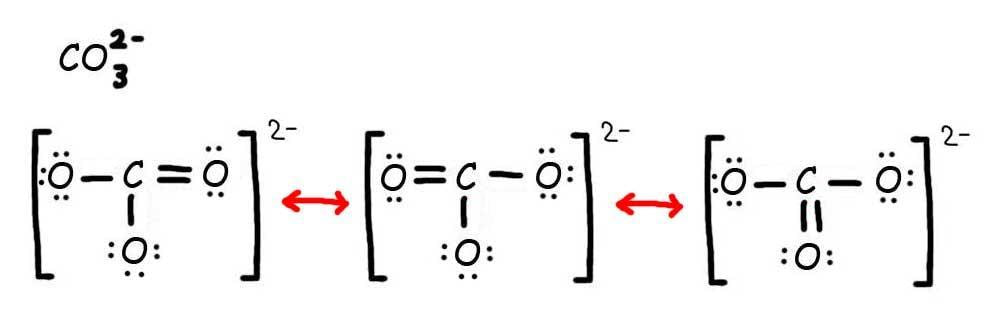
How to Draw Lewis Structures
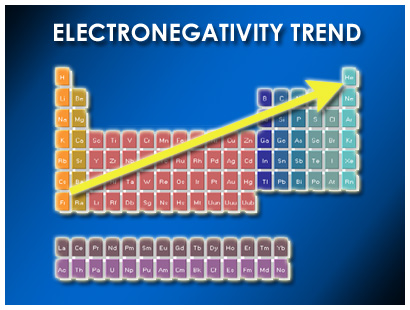
Fluorine has the highest electronegativity:
Ionic Bonding |
Covalent Bonding |
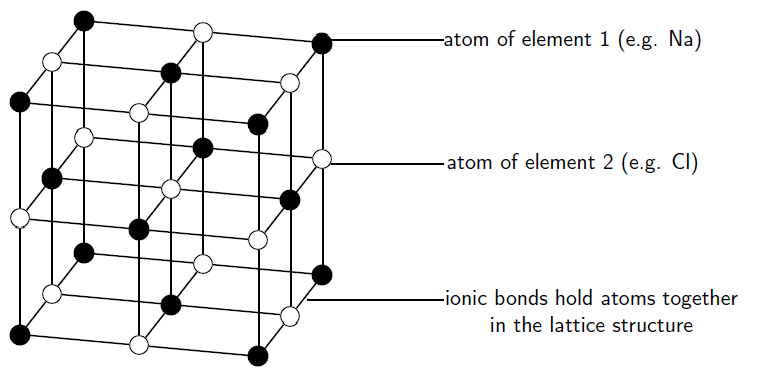
Crystal Structure of the Ionic Compound, Sodium Chloride
|
The History of Poisoned Alcohol Includes an Unlikely Culprit: The U.S. Government - TIME Magazine (January 14, 2015)
History tells of a time when not being able to tell the difference between ethanol and methanol could be deadly.
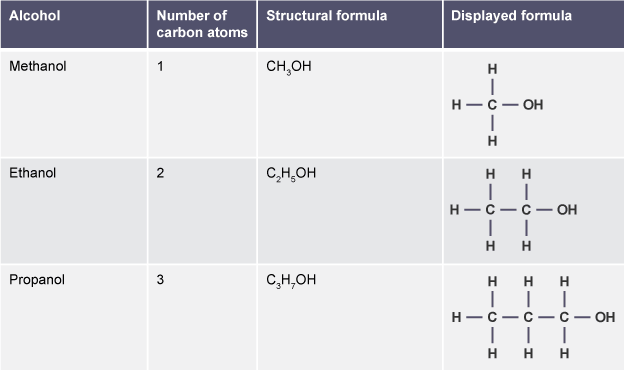
The Lewis Structures for a few alcohols are shown to the right. |
Alcohol Lewis Structure Comparison
|