Unit 4 - Introduction to Solutions
To be in equilibrium is to be in a state of balance. A tug of war, in which the two sides pull with equal force so that the rope does not move, is an example of a static equilibrium, one in which an object is at rest. Equilibria can also be dynamic, whereby a forward process and the reverse process take place at the same rate so that no net change occurs. ~ Chemistry - The Central Science
|
Videos
Solutions
4.1 Video: Solutions ~Bozeman (9:47)
A solution can be either a solid, liquid or gas but must be homogeneous in nature. The solutes can not be separated with a filter and so either chromatography or distillation must be used. Molarity is the number of moles of solute in a Liter of total solution. A simple molar solution preparation is also included. 4.2 Molarity Practice Problems ~DeWitt (9:42)
Confused about molarity? Don't be! Here, we'll do practice problems with molarity, calculating the moles and liters to find the molar concentration. We'll also have to use conversion factors to convert between grams and moles, and between milliliters and liters. Beer's Law
Light & Matter ~Bozeman (8:19)
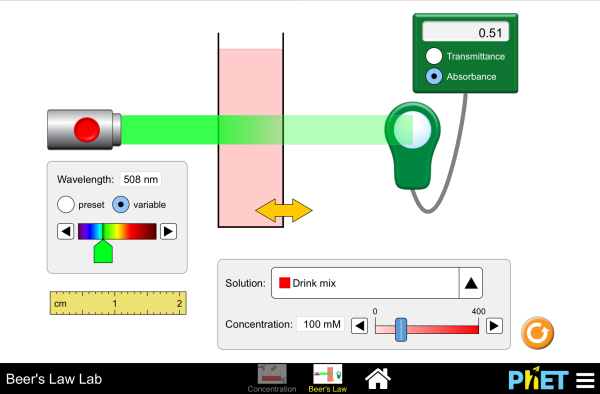
Paul Andersen explains why light is important in probing matter. Beer-Lambert Law describes how increasing concentration increases the absorbance of a solution. [here] AP Chemistry Investigation #1: Use Beer's Law to Determine Blue Dye Concentration in a Sports Drink (16:44) ~Aaron Ulle [here]
Solubility
Solubility Rules
Linear Equations
Solubility Graphs
Activity Series
Net Ionic Equations
Phase Changes
Phase Diagrams - Brightstorm (6:09) Includes a DEMO of CO2 Triple Point
|
Practice/Worksheets/Games
Balancing Chemical Equations
Balancing Chemical Equations
Gapfill Exercise from Science Geek [here] Graphing & Linear Equations
Predictions Involving Single & Double Replacement Reactions
Does This Reaction Happen?
Decide if the reaction actually occurs. You will need an activity series for the single replacement reactions, and a solubility table for the double replacement reactions. Digital Practice [here] Predicting Reaction Products with Answers Worksheet:
[Predicting Reaction Products s/answers] Double Replacement Reactions Practice Worksheet with Answers
Science Geek Link Double Replacement Reactions Practice Worksheet with Answers
Direct Download Type/Balance/Stoichiometry Bundle
Chemical Reactions & Stoichiometry (Simulation) AACT Classify each chemical reaction, then balance the equation and solve a stoichiometry question based on it. [here]
Demonstrations
Electrolytes Activity with Vernier Probes
|
Other Resources
From Our Classroom:
Graphing With Microsoft Excel
Unit 4 Labs
Preparing a Solution Lab
Calculate the requested volume of a specific concentration and prepare the solution correctly.
Beer’s Law Lab
PhET Beer’s Law
Determining the Concentration of a Solution Using Spectrovis & Laptop Computer ~Flinn Scientific (12:39) [here]
Beer’s Law Lab Procedure [here]
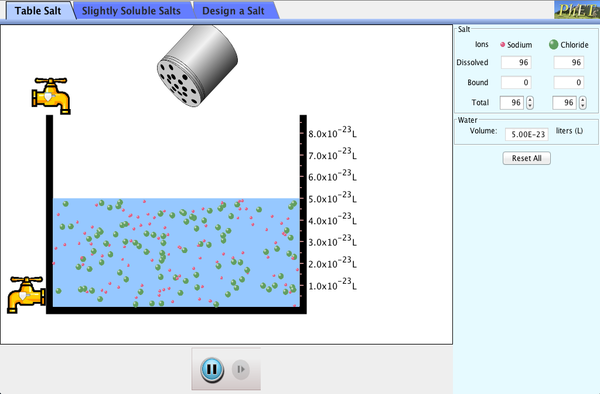
Salts & Solubility
PhET Simulation Salts & Solubility
Making Solutions & Precipitates Lab
Answers to Selected Practice
WS 4.2 Molarity of Solutions
WS 4.3 Chemistry: Molarity & Stoichiometry
WS 4.4 Making Solutions Practice
4.6 Solutions Review
4.8 Solubility Curves
4.11 Writing Net Ionic Equations
Pre-Lab Questions: Beer's Law Lab
Test 4 Review
|
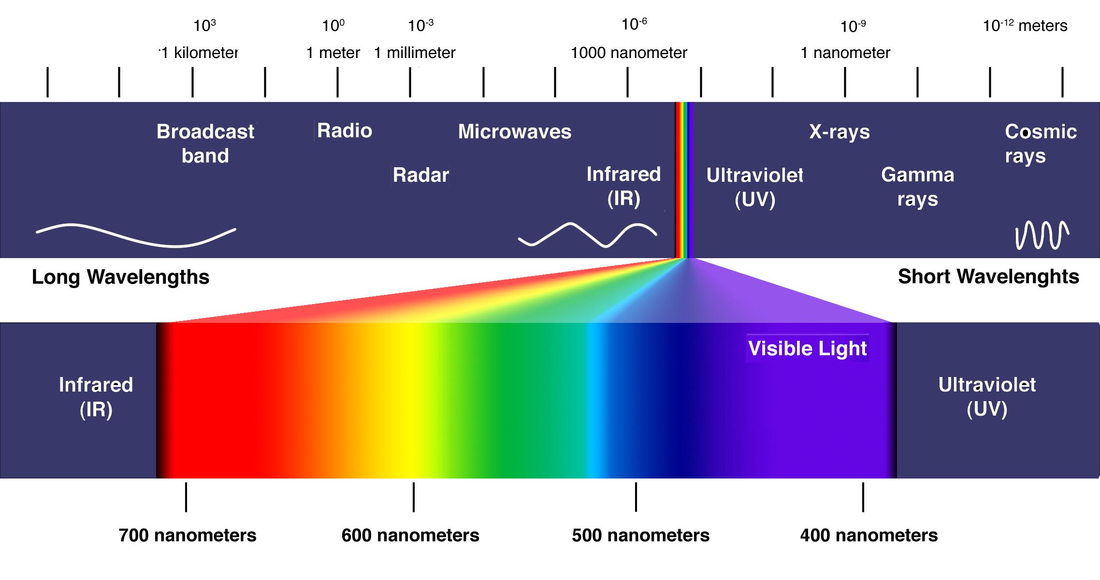
Electromagnetic Spectrum with large takeout for Wavelengths for Visible Light
Spectrometry
The observation and measurement of wavelengths of light or other electromagnetic radiation.
If you arrange some colors in a circle, you get a "color wheel". The diagram shows one possible version of this. An internet search will throw up many different versions!
Colors directly opposite each other on the color wheel are said to be complementary colors. Blue and yellow are complementary colors; red and cyan are complementary; and so are green and magenta. Mixing together two complementary colors of light will give you white light.
What this all means is that if a particular color is absorbed from white light, what your eye detects by mixing up all the other wavelengths of light is its complementary color.
Colors directly opposite each other on the color wheel are said to be complementary colors. Blue and yellow are complementary colors; red and cyan are complementary; and so are green and magenta. Mixing together two complementary colors of light will give you white light.
What this all means is that if a particular color is absorbed from white light, what your eye detects by mixing up all the other wavelengths of light is its complementary color.
Do Wireless Devices Cause Cancer? (Aug 3, 2021) A surge in the number of wireless devices has some researchers concerned that the daily bombardment of electromagnetic signals may increase our cancer risk. Read the article here.
Double-Replacement Reactions
Silver Nitrate in Salt Water
How to Write Net Ionic Equations
Ionic equations and net ionic equations are usually written only for reactions that occur in solution and are an attempt to show how the ions present are reacting. While ionic equations show all of the substances present in solution, a net ionic equation shows only those that are changed during the course of the reaction.
To write ionic equations follow the steps below. Each step will be demonstrated using the reaction of magnesium metal with hydrochloric acid as an example.
1. Write the molecular equation and balance it.
Mg + 2 HCl --> MgCl2 + H2
2. Determine the state of each substance (gas, liquid, solid, aqueous). Use the solubility rules to determine which of the ionic compounds are soluble in water. Soluble ionics are identified with an (aq), insoluble ones with an (s). Most elements and covalent compounds (covalent compounds are formed when two or more nonmetallic elements are bonded to each other) are insoluble in water and should be shown with an (s), (l) or (g).
Mg(s) + 2 HCl(aq) --> MgCl2(aq) + H2(g)
3. Write the ionic equation by breaking all the soluble ionic compounds (those marked with an (aq)) into their respective ions. Each ion should be shown with its charge and an (aq) to show that it is present in solution. Use coefficients to show the number of each ion present. Rewrite the elements and covalent compounds as they appeared in the preceding step.
Mg(s) + 2 H+(aq) + 2 Cl-(aq) --> Mg2+(aq) + 2 Cl-(aq) + H2(g)
4. Write the net ionic equation by removing the spectator ions. Spectator ions are those ions that appear exactly the same on each side of the ionic equation.
Mg(s) + 2 H+(aq) --> Mg2+(aq) + H2(g)
Ionic equations and net ionic equations are usually written only for reactions that occur in solution and are an attempt to show how the ions present are reacting. While ionic equations show all of the substances present in solution, a net ionic equation shows only those that are changed during the course of the reaction.
To write ionic equations follow the steps below. Each step will be demonstrated using the reaction of magnesium metal with hydrochloric acid as an example.
1. Write the molecular equation and balance it.
Mg + 2 HCl --> MgCl2 + H2
2. Determine the state of each substance (gas, liquid, solid, aqueous). Use the solubility rules to determine which of the ionic compounds are soluble in water. Soluble ionics are identified with an (aq), insoluble ones with an (s). Most elements and covalent compounds (covalent compounds are formed when two or more nonmetallic elements are bonded to each other) are insoluble in water and should be shown with an (s), (l) or (g).
Mg(s) + 2 HCl(aq) --> MgCl2(aq) + H2(g)
3. Write the ionic equation by breaking all the soluble ionic compounds (those marked with an (aq)) into their respective ions. Each ion should be shown with its charge and an (aq) to show that it is present in solution. Use coefficients to show the number of each ion present. Rewrite the elements and covalent compounds as they appeared in the preceding step.
Mg(s) + 2 H+(aq) + 2 Cl-(aq) --> Mg2+(aq) + 2 Cl-(aq) + H2(g)
4. Write the net ionic equation by removing the spectator ions. Spectator ions are those ions that appear exactly the same on each side of the ionic equation.
Mg(s) + 2 H+(aq) --> Mg2+(aq) + H2(g)
Credits
viper and mouse pictures credit: Nature News Online Journal
Complementary Color Wheel: Rez to the City Blog
Complementary Color Explanation: Chemistry LibreText