Unit 12 Chemical Kinetics
Big Idea 4: The rates of chemical reactions are determined by molecular collisions.
Check this out first:
How to Learn Chemistry & a Lesson on Collision Theory ~ Michael Farabaugh (6:43)
Helpful advice for learning chemistry, and a talk about collision theory, which relates to chemical kinetics and reaction rates. [here]
Helpful advice for learning chemistry, and a talk about collision theory, which relates to chemical kinetics and reaction rates. [here]
The study of the rates of chemical reactions and the factors that influence them is known as chemical kinetics.
The Chemical Kinetics unit addresses four methods of determining rate:
- Instantaneous Rate
- Relative Rate
- Differential Rate Law
- Integrated Rate Law
Reaction Rates
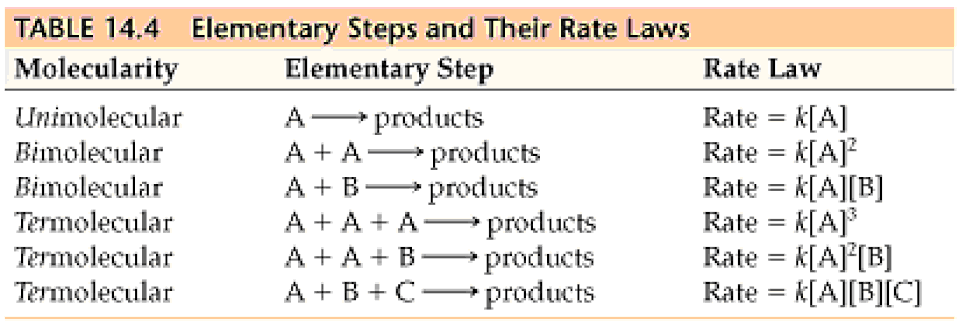
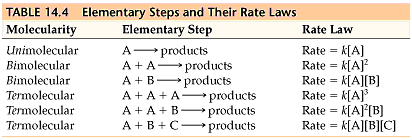
Rate Laws
- There are two types of rate laws.
- The differential rate law (often called simply the rate law) shows how the rate of a reaction depends on concentrations.
- The integrated rate law shows how the concentrations of species in the reaction depend on time.
- Because we typically consider reactions only under conditions where the reverse reaction is unimportant, our rate laws will involve only concentrations of reactants.
- Because the differential and integrated rate laws for a given reaction are related in a well-defined way, the experimental determination of either of the rate laws is sufficient.
- Experimental convenience usually dictates which type of rate law is determined experimentally.
- Knowing the rate law for a reaction is important mainly because we can usually infer the individual steps involved in the reaction from the specific form of the rate law.
Determining the Form of the Rate Law
|
Videos
|
|
Bozeman AP Videos
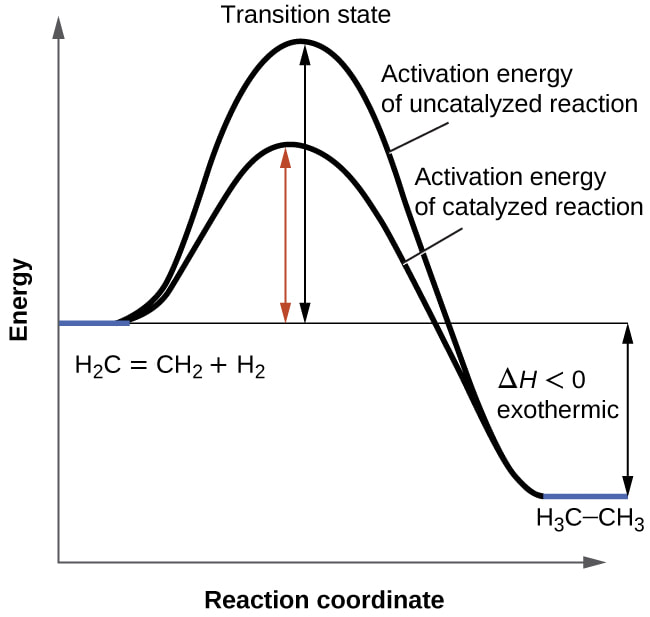
Catalyzed vs Uncatalyzed Rxn @OpenTextBC
|
Dr. Leggett Videos
|
Other Resources
Forest Classroom Resources:
Notes
Summaries to Help Review Kinetics:
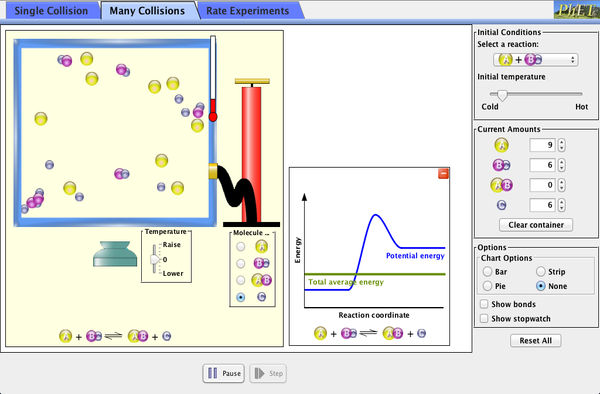
Simulations
Click to set custom HTML
|
Vocabulary
spontaneity - refers to the inherent tendency for the process to occur; however, it implies nothing about speed.
chemical kinetics - the area of chemistry concerned with reaction rates.
reaction mechanism - the series of steps by which a reaction takes place.
spontaneity - refers to the inherent tendency for the process to occur; however, it implies nothing about speed.
chemical kinetics - the area of chemistry concerned with reaction rates.
reaction mechanism - the series of steps by which a reaction takes place.
Integrated first-order rate law ln[A] = - kt + ln[A]0