Unit 2 - Compound Structure & PropertiesIntermolecular forces are physical forces between molecules. They explain how molecules interact with each other. The strength or weakness of intermolecular forces determine the state of matter of a substance (e.g., solid, liquid, gas) and some of the chemical properties (e.g., melting point, structure). There are three major types of intermolecular forces: London dispersion force, dipole-dipole interaction, and ion-dipole interaction.
The energy required to break molecules apart is much smaller than a typical bond-energy. Intermolecular Forces are particularly important in terms how molecules interact and form biological organisms or even life. This link gives an excellent introduction to the interactions between molecules. ~
|
|
|
See Some Examples
|
The lotus plant grows in aquatic environments. The surface is highly water repellent. “Superhydrophobic” leaf causes any water to bead up and roll off. Water collects dirt, keeping the plant clean. This self-cleaning property is why the lotus plant is considered a symbol of purity in many Eastern cultures.
|
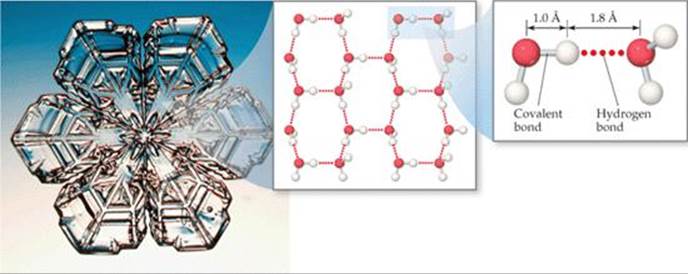
Snowflakes grow from water vapor around some kind of nucleus, such as a bit of dust.
|
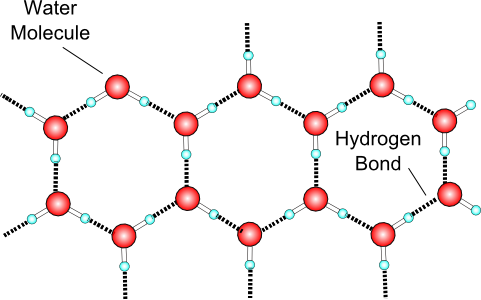
As more water molecules stick together with hydrogen bonds, they form a regular pattern, as shown here.
The reason these patterns form in snowflakes is that the water molecules have a V shape, and hydrogen bonds form at the ends of these Vs. |
Intermolecular Forces Explain “Like Dissolves Like” (Warning: Do not use “Like Dissolves Like” as an explanation on the test.)
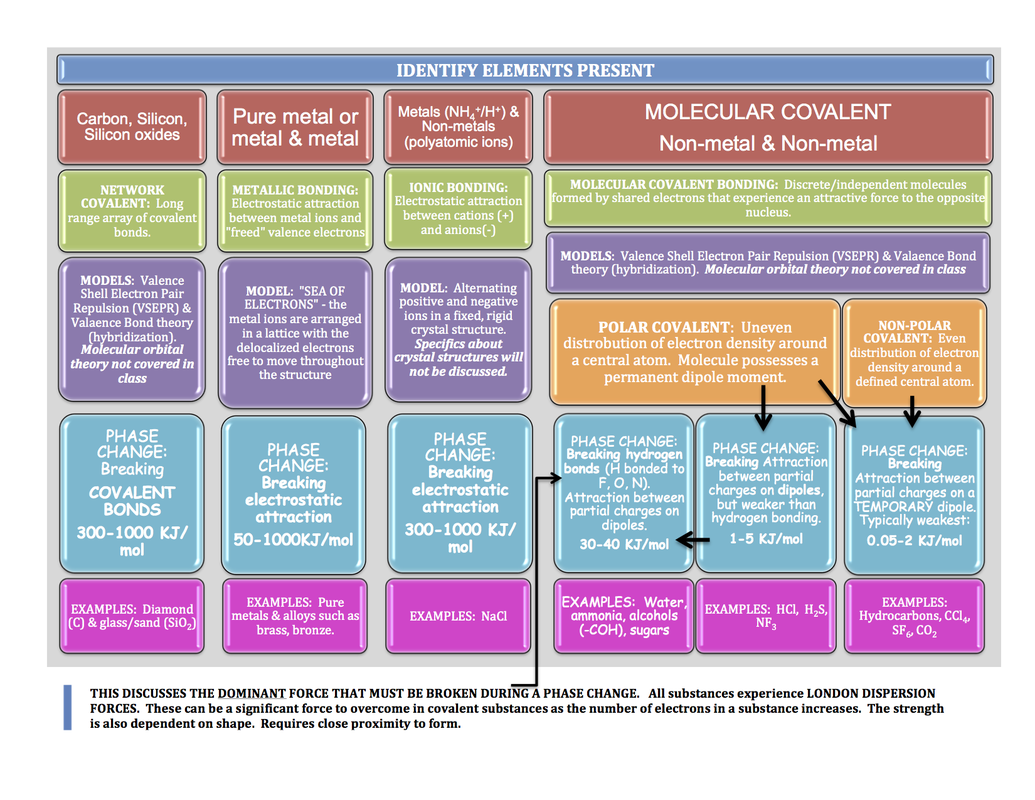
Intermolecular forces are noncovalent forces between molecules. There are a variety of these forces but the most common are: dipole-dipole interactions, hydrogen bonding and van der Waals (or London) forces. Of these, hydrogen bonds are the strongest followed by dipole-dipole interactions and finally, van der Waals forces. Thus a sample of a substance capable of hydrogen bonding will have stronger forces between its molecules than those between the molecules of a substance which is only capable of van der Waals forces. This means that it will take more energy to disrupt those forces between molecules and the first substance should have the higher boiling and melting points.
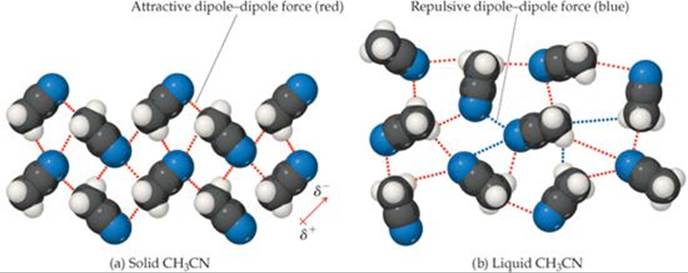
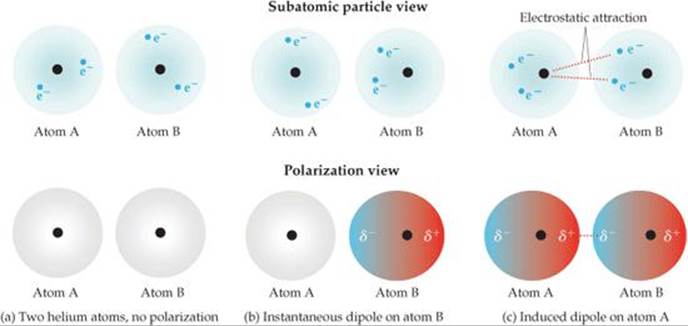
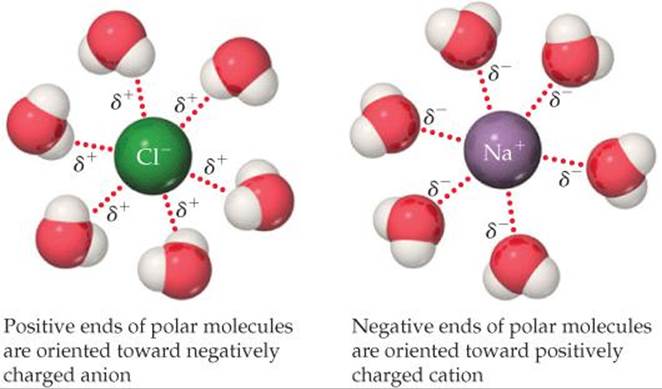
How do we know which type of intermolecular forces exist between the molecules of a given substance? You must be able to figure out whether or not the molecules are polar (see the Molecular Polarity module). If the molecule is polar, it has areas of partial positive and partial negative charges which can interact with the areas of partial negative and partial positive charges in a second molecule (since opposites are attracted to each other). This type of force is called a dipole-dipole interaction. Hydrogen bonds are a special type of dipole-dipole interaction but are about five times stronger and thus are considered to be a distinct intermolecular force. Hydrogen bonds also involve the attraction of a partial positive atom in one molecule to a partial negative atom in a second molecule (and vice versa) but in this case the partial positive is a hydrogen atom which is VERY partial positive by virtue of the fact that it is covalently bonded to a small, electronegative atom (F, Cl, O, N). This VERY partial positive hydrogen atom will be VERY strongly attracted to the partial negative atom in the second molecule thus making the strongest intermolecular force, the hydrogen bond. If the molecule is nonpolar, then no dipole-dipole interactions or hydrogen bonding can occur and the only possible intermolecular force is the weak van der Waals force. This force is sometimes called the induced dipole-induced dipole interaction since it involves the transient creation of an induced dipole within a nonpolar molecule due to the close proximity of the electron cloud of a second molecule. As soon as the second molecule moves away, the induced dipole disappears. While the induced dipole exists, that portion of the molecule is attracted to other molecules but as soon as the induced dipole disappears, the attraction also disappears. Thus this type of force is weak and short-lived and occurs between nonpolar molecules.
Having been reminded of the three major types of intermolecular forces, you can now understand the solvent rule, "like dissolves like". If you have a polar solute, you will want to choose a polar solvent so that the solvent can solvate (surround and form dipole-dipole interactions or hydrogen bonds) the solute molecules. If you choose a nonpolar solvent, the polar solute molecules will be attracted to each other and not to the solvent molecules and no solvation will occur. If, however, you have a nonpolar solute, you will want to choose a nonpolar solvent so that the solute and solvent molecules can interact by van der Waals interactions. If you choose a polar solvent, the solvent molecules will be attracted to each other and not to the solute molecules and no solvation will occur. In order for solvation to occur and a stable solution to exist, the solute and solvent molecules must be able to form intermolecular associations. For this to occur, the solute and solvent molecules must be similar (both polar or both nonpolar); i.e., they must be "alike".
Taken from Armstrong Chemistry/Physics
How do we know which type of intermolecular forces exist between the molecules of a given substance? You must be able to figure out whether or not the molecules are polar (see the Molecular Polarity module). If the molecule is polar, it has areas of partial positive and partial negative charges which can interact with the areas of partial negative and partial positive charges in a second molecule (since opposites are attracted to each other). This type of force is called a dipole-dipole interaction. Hydrogen bonds are a special type of dipole-dipole interaction but are about five times stronger and thus are considered to be a distinct intermolecular force. Hydrogen bonds also involve the attraction of a partial positive atom in one molecule to a partial negative atom in a second molecule (and vice versa) but in this case the partial positive is a hydrogen atom which is VERY partial positive by virtue of the fact that it is covalently bonded to a small, electronegative atom (F, Cl, O, N). This VERY partial positive hydrogen atom will be VERY strongly attracted to the partial negative atom in the second molecule thus making the strongest intermolecular force, the hydrogen bond. If the molecule is nonpolar, then no dipole-dipole interactions or hydrogen bonding can occur and the only possible intermolecular force is the weak van der Waals force. This force is sometimes called the induced dipole-induced dipole interaction since it involves the transient creation of an induced dipole within a nonpolar molecule due to the close proximity of the electron cloud of a second molecule. As soon as the second molecule moves away, the induced dipole disappears. While the induced dipole exists, that portion of the molecule is attracted to other molecules but as soon as the induced dipole disappears, the attraction also disappears. Thus this type of force is weak and short-lived and occurs between nonpolar molecules.
Having been reminded of the three major types of intermolecular forces, you can now understand the solvent rule, "like dissolves like". If you have a polar solute, you will want to choose a polar solvent so that the solvent can solvate (surround and form dipole-dipole interactions or hydrogen bonds) the solute molecules. If you choose a nonpolar solvent, the polar solute molecules will be attracted to each other and not to the solvent molecules and no solvation will occur. If, however, you have a nonpolar solute, you will want to choose a nonpolar solvent so that the solute and solvent molecules can interact by van der Waals interactions. If you choose a polar solvent, the solvent molecules will be attracted to each other and not to the solute molecules and no solvation will occur. In order for solvation to occur and a stable solution to exist, the solute and solvent molecules must be able to form intermolecular associations. For this to occur, the solute and solvent molecules must be similar (both polar or both nonpolar); i.e., they must be "alike".
Taken from Armstrong Chemistry/Physics
|
Videos
|
Practice/Games/Quizzes
|
Resources
|
|
018-Intermolecular Forces ~Bozeman Science (7:00)
How intermolecular forces differ from intramolecular forces. How differences in these forces account for different properties in solid, liquids and gases. Some of these properties include the boiling point, melting point, surface tension, capillary action and miscibility. Hydrogen Bonding & Common Mistakes ~Tyler DeWitt (8:59)
Hydrogen bonding can be so confusing. Hydrogen bonds are intermolecular forces between molecules. They form because one atom has a high electronegativity, so it gets a partial negative charge, and the hydrogen gets a partial positive charge. |
Water, Polarity & Hydrogen Bonds (Interactive Tutorial) ~ScienceMusicVideos.com
Polar Water vs Less Polar Alcohol: A Virtual Lab ~Science MusicVideos.com
|
Resources from Class
|
|
Polar Bonds, Polarity & Intermolecular Forces ~Mr. Causey (9:57)
Mr. Causey discusses intermolecular forces (IMF), polarity, polar bonds and polar molecules. Why is Water "STICKY"?
Water: Chemistry & Properties, Part 1 (4:25) |